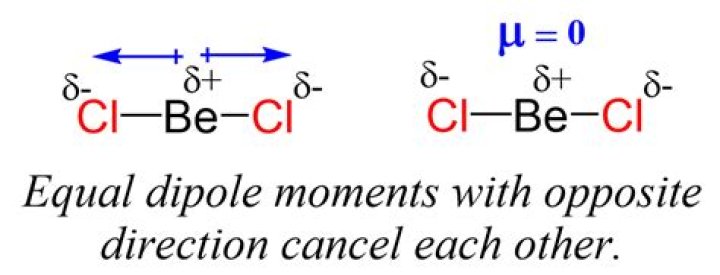
This means there is no net dipole in the molecule, thus BeCl2 has no dipole moment..
Likewise, does BeCl2 have a dipole?
Answer: Because the polarities cancel each other. The electronegativity of chlorine is greater than beryllium and thus both the bonds are polar but as the shape is linear, the polarities cancel each other as they are equal and opposite and the molecule becomes non polar.
Similarly, is BeCl2 polar or nonpolar? If the pull from each of the bonds cancels each other out, like forces that are equal in magnitude but opposite in direction, the molecule will be nonpolar. Examples of this are BeCl2, AlCl3 and CF4. If the pull from the polar bonds in a molecule do not cancel each other out, the molecule is polar.
Also to know is, does BeCl2 have net dipole moment?
In BeCl2, the Cl atom is more electronegative than Beryllium atom so the direction of dipole moment vector is towards chlorine atom. All the Be-Cl polar bonds canceled each other's polarity with net dipole moment is zero.
What is dipole moment BeCl2?
BeCl2 is sp hypridized. It is a linear molecule; therefore, the dipole moment is zero.
Related Question Answers
Why is BeCl2 nonpolar?
BeCl2 is nonpolar. Beryllium chloride or BeCl2 has a linear electron geometry with no lone pairs, making it nonpolar because of its Lewis structure and VSEPR model. BeCl2 is not an ion because the electronegativity difference between Cl and Be atoms are not sufficient to form an ionic bond.Is PCl3 dipole dipole?
(a) PCl3 is polar while PCl5 is nonpolar. As such, the only intermolecular forces active in PCl5 are induced dipole-induced dipole forces (London dispersion forces). In PCl3, there are also dipole-dipole forces and dipole-induced dipole forces.Why is the dipole moment of bf3 zero?
Boron trifluoride is has zero dipole moment although it has three polar B—F bonds. This is because BF3 has sp2 hybridisation and regular trigonal planal geometry. And hence the individual dipole moments of polar bonds get cancelled and overall dipole moment is zero.What does dipole moment mean?
A dipole moment is a measurement of the separation of two opposite electrical charges. Dipole moments are a vector quantity. The magnitude is equal to the charge multiplied by the distance between the charges and the direction is from negative charge to positive charge: μ = q · r.Why is SCl2 polar?
Is SCl2 polar or non-polar? As people say, Cl-S-Cl is polar because it is “bent” with a bond angle close to a right angle. Sigma overlap of one chlorine 3p orbital with the sulfur 3py and another with the 3pz orbital gives an unusual valency of 2 for sulfur and bonds at almost right angles.Does nh3 have a dipole moment?
NH3 has a dipole moment while BF3 does not. By the VSEPR theory, the shape of the NH3 molecule turns out to be tetrahedral with 3 bond pairs and 1 lone pair. Thus, there is no chance of cancellation of the dipole vector as the vectors are inclined at an angle of 107°.Does H2Se have a dipole moment?
H2S, H2Se and H2Te exhibit dipole-dipole intermolecular forces while H2O exhibits hydrogen bonding. CO2 is a nonpolar molecule (vectors cancel), so it only has the dispersion force (22 electrons) and has the weakest attractive force of the four (bp −78°C). Hg is a metallic solid.Does xef4 have a dipole moment?
Dipoles and Electrostatic Surfaces XeF4, ClF3 and CCl3Br The Xe-F bonds are all polarized but they cancel one another out so the molecule has no dipole. Chlorine trifluoride has three polarized bonds and they combine to produce a small molecular dipole along the Cl-F bond.What is the dipole moment of bacl2?
why bacl2 has 0 dipole moment ? BaCl2 (in gas phase) has zero dipole moment even though the bonds are polarised. The structure of BaCl2 is linear that is Cl-Ba-Cl. The chloride pull the electrons toward them, but one of the chloride pulling it to the right and other pulling it to the left.Why does BCl3 not have a dipole moment?
B-Cl has a dipole due to the difference in the elctronegativity of boron and chlorine atom. The overall dipole of a molecule also depends on the geometry. The geometry of BCl3 is planar with a bond angle of 120 degree. The resultant dipole of two B-Cl bonds cancels the third one, resulting in net zero dipole.Is HCL polar or nonpolar?
HCL is a polar molecule as chlorine has a higher electronegativity than the hydrogen. Thus, it attracts electrons to spend more time at its end, giving it a negative charge and hydrogen a positive charge. How do you know if Br2 is polar or nonpolar?Is water polar or nonpolar?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This is an example of polar covalent chemical bonding.Is h2o molecule polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.Why is XeF2 nonpolar?
According to the VSEPR theory, The molecular geometry of the molecule is linear. As there are fluorine molecules on both the side of the central atom, there is no dipole moment and hence there is no polarity. XeF2 is nonpolar due to the symmetric arrangement of the bonded pairs of electrons.Why is CH3Cl polar?
Since the H is between B and C in terms on electronegativity values, their difference in electronegativity values is so small, the C-H bond is considered nonpolar; thus, no dipole arrow is drawn for the C-H bonds. Because the C-Cl bond is polar, the CH3Cl has a net dipole, so CH3Cl is polar.Is NaCl a polar molecule?
Yes, NaCl is an ionic bond which makes it polar. A polar molecule consists of atoms having either a positive or negative bond. In this case, Na has a +1 charge and Cl has a -1 charge, making the bond polar. The difference in electronegativities is what makes a bond polar or nonpolar.Is BCl3 polar?
So BCl3 is a non polar molecule yes and the bonds are polar so why is this? Well the reason behind is the electronegativity of elements. We can see that Cl has electronegativity of 3.16 and B 2.04. Now imagine you make a single bond between Cl and B what you get is polar bond and a polar molecule.How many lone pairs are in BeCl2?
three lone pairs
What is the bond order of N 2?
Answer and Explanation: In a N2+ molecule , the bond order is 2.5. In a regular N2 molecule, there are 10 total valence shell electrons present in the molecular orbitals.