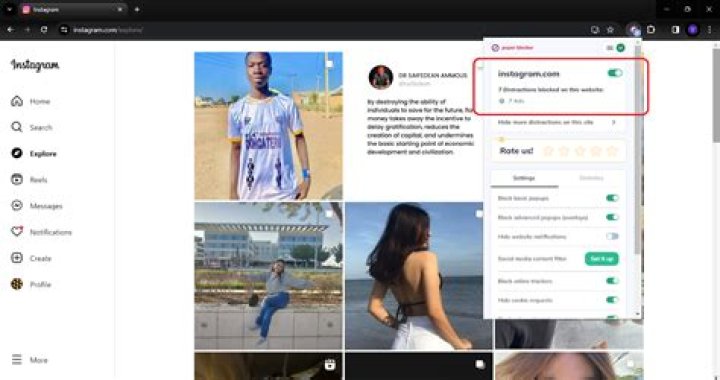
Steam is water in the gas phase. It is commonly formed by boiling or evaporating water. If liquid water comes in contact with a very hot surface or depressurizes quickly below its vapor pressure, it can create a steam explosion..
In respect to this, how is steam produced in a boiler?
A boiler incorporates a firebox or furnace in order to burn the fuel and generate heat. The generated heat is transferred to water to make steam, the process of boiling. This produces saturated steam at a rate which can vary according to the pressure above the boiling water.
Additionally, what causes wet steam? When a steam boiler heats up water, bubbles breaking through the water surface will pull tiny water droplets in with the steam. Unless a superheater is used, this will cause the steam supply to become partially wet (wet steam) from the added liquid.
Also know, does Steam create pressure?
The reason steam creates pressure is that steam is a gas, and gases always expand to fill the available space. If the gas is within a flexible container, like a balloon, the gas will push outwards on the sides of the container and make it expand.
Is Steam a liquid or a gas?
Steam is water in the gas phase, which is formed when water boils or evaporates. Steam is invisible; however, "steam" often refers to wet steam, the visible mist or aerosol of water droplets formed as this water vapour condenses.
Related Question Answers
What is the function of steam?
To produce steam for various purposes (hence, the name steam plant) Steam can be used to generate electricity, to be used as a form of pre heating the heat exchanger if required..or it can be used to drive a pump like most oil and gas industries..What are the applications of steam?
Steam is used in a wide range of industries. Common applications for steam are, for example, steam heated processes in plants and factories and steam driven turbines in electric power plants, but the uses of steam in industry extend far beyond this.What is boiler principle?
The basic working principle of boiler is as follow: The boiler is essentially a closed vessel inside which water is stored. Fuel (generally coal) is burnt in a furnace and hot gasses are produced. Thus, the basic working principle of boiler is to convert water into steam by using heat energy as input.What is steam test in boiler?
Accumulation test of the boiler. A boiler test to ensure that the safety valves can release steam fast enough to prevent the pressure rising by 10%. The main steam stop valve is closed during the test.How many types of steam are there?
Now, coming to the types of steam there are basically three types of steam based on their states which are as follows: Wet steam (Saturated steam) Dry steam (Dry saturated steam) Superheated steam.What is the capacity of a boiler?
Galvanized Steel Industrial Steam Boiler, Capacity (kg/hr): 500-1000 kg/hr
| Capacity (kg/hr) | 500-1000 kg/hr |
| Working Pressure | 5-10 kg/cm2g |
| Material | Galvanized Steel |
| Minimum Order Quantity | 1 Set |
What is steam and types?
Type of Steam. Pressure –Temperature Relationship of Water & Steam: If water is heated beyond the boiling point, it vaporizes into steam, or water in the gaseous state. Saturated (dry) steam results when water is heated to the boiling point (sensible heating) and then vaporized with additional heat (latent heating).What is boiler efficiency?
Boiler efficiency is a measure of how effectively chemical energy in fuel is converted into heat energy in steam going to the turbines.Is Steam pure water?
What is distilled water? Distilled water is also called steam-distilled water. Distilled water is water which has been heated to the boiling point so that impurities are separated from the water, which becomes vapor or steam at 212 degrees farenheit(100C.). Pure water contains no solids, minerals, or trace elements.What is the maximum temperature of steam?
Steam cycle parameters with a main steam pressure above 250 bar and temperature above 565 °C and up to 600 °C (main steam) and 620 °C (reheat) are commonly called ultra supercritical (USC); these higher steam conditions can be achieved because of the development of new ferritic materials.Is Steam always water?
Steam is water in the gas phase. It is commonly formed by boiling or evaporating water. Steam that is saturated or superheated is invisible; however, "steam" often refers to wet steam, the visible mist or aerosol of water droplets formed as water vapour condenses.What is the pressure of steam?
If your mill has a boiler producing saturated steam at, say, 250 psi and you are using steam at your dry kilns at 100 psi (which has been reduced from 250 psi through a pressure reducing valve), the steam will be 407°F, not at the saturation temperature of 338°F for 100 psi steam.Is steam hotter than water?
Steam occurs when water goes above 212 degrees Fahrenheit, which is hotter than water when it is at its stable point. As a result, burns from steam that is coming from a pot of boiling water because it is at a higher temperature than the water itself.Why is steam so powerful?
The water is still nearby, but it's now in a gaseous form called steam. This form of water is also called water vapor, and it's very powerful stuff. This is because steam has a lot of energy. This is because as you continue to add more heat, more water molecules turn to vapor, and then you're not heating them anymore!What is the density of steam?
"At 100 ℃ and atmospheric pressure the heat of vaporization of steam is 2260 kJ/kg, the density of water is 103 kg/m3, and the density of steam is 0.6 kg/m3."What is the enthalpy of steam?
Total enthalpy in saturated steam at atmospheric pressure (hg) = 2 675 kJ/kg. Therefore, at the lower pressure state of 0 bar g. Total enthalpy in the water = 0.888 kg x 419 kJ / kg = 372 kJ (A) Total enthalpy in the steam = 0.112 kg x 2 675 kJ / kg = 299 kJ (B)What is the temperature of steam?
When water is heated at atmospheric pressure, its temperature rises until it reaches 212°F, the highest temperature at which water can exist at this pressure. Additional heat does not raise the temperature, but converts the water to steam.What is the difference between wet and dry steam?
Wet steam is a mixture of steam and liquid water. It is said to be a two-phase mix: steam contains droplets of water that have not changed phase. Dry steam is steam that is at the temperature of saturation, but does not contain water particles in suspension. It has a very high dryness fraction, with almost no moisture.What is the quality of steam?
Steam quality is the proportion of saturated steam (vapor) in a saturated condensate (liquid)/ steam (vapor) mixture. A steam quality of 0 indicates 100 % liquid, (condensate) while a steam quality of 100 indicates 100 % steam.