How many valence electrons does ClO2?
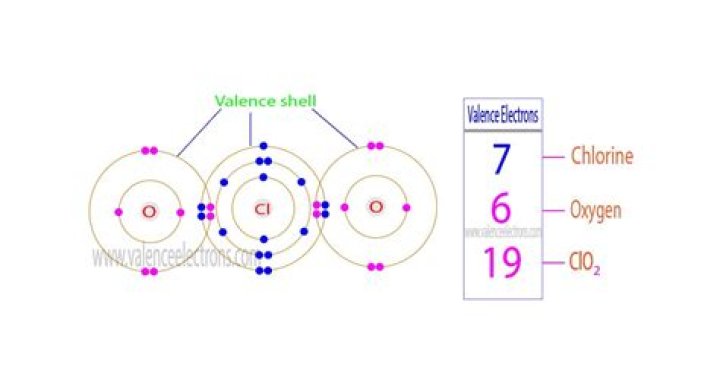
.
Moreover, how many valence electrons does chlorite have?
The valence electrons you have availableare: 1 Cl + 2 O + 1 e=1×7+2×6+1=20.
what is the molecular shape of ClO2 -? Re: Bond angle of ClO2- In doing so, the electron pair geometry of themolecule is tetrahedral and the molecular geometry isbent. Since there are two lone pairs on chlorine, the electron pairrepulsion will result in a bond angle that is slightly less than109.5.
Correspondingly, how many unshared electrons does ClO2 have?
The ClO2 Lewis structure has 19 valenceelectrons meaning that there will be an odd number ofvalence electrons in the structure. For the Lewis structurefor ClO2 you should take formal charges into account to findthe best Lewis structure for the molecule.
How many covalent bonds are in ClO2?
Under acidic conditions; CLO2 results in aneutral molecule consisting of three electronegative atoms heldtogether by covalent and coordinate covalentbonds.
Related Question AnswersWhat is the total number of valence electrons in the Lewis structure of co2?
CO2 Lewis structure So CO2 = 4 + 6(2) = 16. So, total valenceelectrons are 16. Carbon is the least electronegative thatmeans it stays at the center. So, put the Carbon in the middle andthen set the oxygen either side of that!How do you determine valence electrons?
For neutral atoms, the number of valenceelectrons is equal to the atom's main group number. The maingroup number for an element can be found from its column on theperiodic table. For example, carbon is in group 4 and has 4valence electrons. Oxygen is in group 6 and has 6 valenceelectrons.How do you find the formal charge?
The formal charge is determined by the valenceelectrons (VE), nonbonding electrons (NE), and bonding electrons(BE) in the following formula: By calculating the formalcharge of each atom in a molecule, we can determine the overallcharge of the molecule.Can chlorine have more than 8 valence electrons?
Re: Having more than 8 valenceelectrons Some molecules can accommodate more than 8valence electrons because they have expandedvalence shells. Instead of being restricted to making4 bonds with 4 chlorine atoms, it can makefive bonds, one for each chlorine atom, using 2electrons for each bond.Is ClO2 a free radical?
Chlorine Dioxide ClO2 is a neutral free radical with C2vsymmetry and its overall dipole moment is 1.792Debye.What is the name for the ion with the formula ClO2?
In chemistry, hypochlorite is an ion composed ofchlorine and oxygen, with the chemical formula ClO−.The chlorite ion, or chlorine dioxide anion, isClO2−.What is the name for ClO2?
Chlorine dioxide is a chemical compound with theformula ClO2.What is hybridization of ClO2?
Hybridisation of ClO2. hybridization. Findthe hybridization as well identify the pπ-pπ as wellas pπ-dπ bonds in ClO2. ClO2 has 2 σ bonds, 1 lonepair, 2π bonds and 1 odd electron. Hybridisation is equalto number of σ bonds + lone pairs.How many pairs of valence electrons are in BrF?
In BrF5, there are 5 bonding pairs ofelectrons and one non-bonding pair ofelectrons.What is ClO2 used for?
Chlorine dioxide gas is used to sterilizemedical and laboratory equipment, surfaces, rooms and tools.Chlorine dioxide can be used as oxidizer ordisinfectant. It is a very strong oxidizer and it effectively killspathogenic microorganisms such as fungi, bacteria andviruses.Why is ClO2 paramagnetic?
Paramagnetism is due to the presence of at least oneunpaired electron in the molecule. Thus, simple compounds such asNO, NO2 nd ClO2 are paramagnetic as the total numbersof electrons in their molecules are odd (NO-15, NO2-23 andClO2 -33 electrons). Though NO2 is paramagnetic, itsdimer, N2O4, is diamagnetic.How do I calculate bond order?
To calculate bond order in chemistry, subtractthe number of the electrons in the antibonding molecules from thenumber of electrons in the bonding molecules. Divide theresult by 2 to get the result. The higher the bond order,the more stable the molecule.What is the structure of Cl2O?
Chlorine monoxide| PubChem CID: | 24646 |
|---|---|
| Structure: | Find Similar Structures |
| Molecular Formula: | ClO or Cl2O |
| Chemical Names: | Chlorine monoxide Dichlorine monoxide Dichlorine oxide CHLORINEOXIDE (CL2O) Cl2O More |
| Molecular Weight: | 86.9 g/mol |