32 valence electrons
.
Also asked, how many valence electrons are in sulfite ion?
Where V = (6 + 6 + 6 + 6 ) – (-2) = 26 , V is the number of valence electrons of the ion. Therefore, the structure in Step 1 is a plausible Lewis structure of SO3-2. Electrons are placed around each atom so that the octet rule is obeyed. Since S is a third row element can accommodate more than 8 valence electrons.
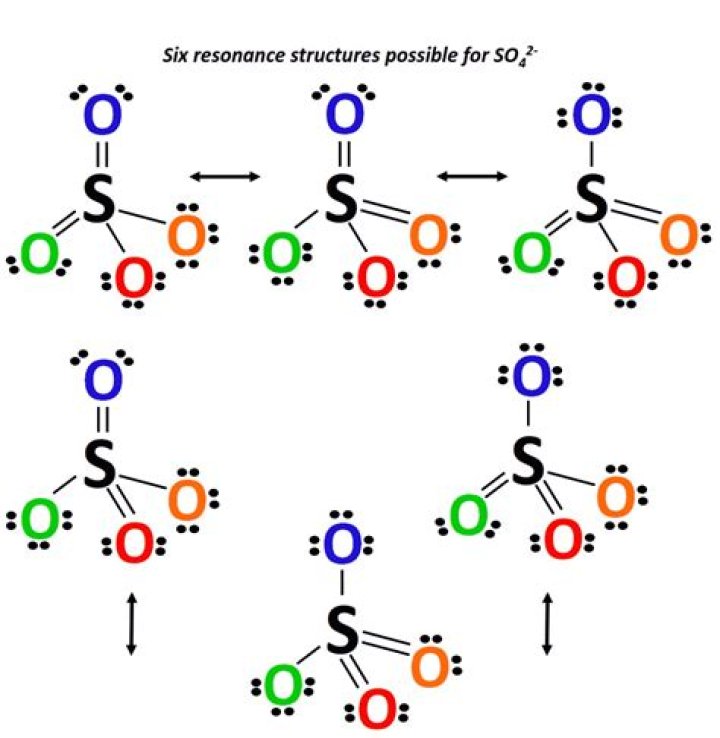
Also, what is the Lewis structure for so42? Let's do the SO4 2- Lewis structure for the sulfate ion. On the periodic table: Sulfur has 6 valence electrons; Oxygen also has 6, we have 4 Oxygens, multiply by 4; and these 2 valence electrons up here, we need to add those, as well. That gives us a total of 32 valence electrons.
Keeping this in view, how many electrons are in a sulfate ion?
For each molecule of sulphate ion, there are 16 electrons with the sulphur, 8 electrons for each oxygen, and 2 additional electrons resulting from the charge. That is a total of electrons.
What is sulfurs charge in so42 -?
An S atom has 6 valence electrons and a O atom has 6 valence elections. SO4 => (6x1) + (6x4) = 30 valence electrons. We would need 32 valence electrons for 4 full outer shells(8x4) but only have 30. Thus the charge is 2- for the ion.
Related Question Answers
How do you find the Valency of so4?
Valency of SO4 = 2 To get valency of SO4, let us consider valency of sulphur and oxygen. Sulphur has 6 electrons in its outer shell. Oxygen has a oxidation no. -2, for four oxygen atoms, we have oxidation number of -8.Is so3 2 polar or nonpolar molecule?
Think of the sulfite ion as a molecule with its geometry and dipole moment AND a net charge. The electron pair geometry is tetrahedral and the molecular geometry is trigonal pyramidal and because of its asymmetrical shape and polar bonds, sulfite has a net dipole moment (2.04D). The ion is polar. The net charge is -2.How many electrons does potassium ion have?
Answer and Explanation: The correct formula, or symbol, for a potassium ion with 18 electrons is K+. A neutral atom of potassium will have 19 electrons to balance theWhat is the shape of so3 2?
trigonal pyramidal
How do you find the formal charge?
Formal Charge = [# valence electrons on neutral atom] – [(# lone electron pairs) + (½ # bonding electrons)] Valence electrons = corresponds to the group number of the periodic table (for representative elements). Lone Pairs = lone electrons sitting on the atom. Each electron counts as one and so a pair counts as two.How many protons and electrons are in a sulfate ion so4 2?
1)How many protons and electrons are in a sulfate ion SO4^2- ? I know the answer is 48 protons and 50 electrons but i do not know how to get this from the ion.What is an octet of electrons?
Usually an “octet of electrons” refers to the outer shell of valence electrons (ones of the highest energy level) being stabilized with 8 valence electrons.What is the atomic number of sulfate?
S-32 is the most common isotope of sulfur. It's atomic number is 16 (number of protons); so, there are 16 neutrons (32–16). The total neutrons for sulfate ion is 16+8+8+8+8 or 48 neutrons.What is sulfate charge?
The sulfate ion carries an overall charge of −2 and it is the conjugate base of the bisulfate (or hydrogen sulfate) ion, HSO − 4, which is in turn the conjugate base of H. 2SO. 4. , sulfuric acid.How many protons and electrons are in sulfide ion?
Thus, each atom or ion of sulfur must contain 16 protons. We are told that the ion also has 16 neutrons, meaning the mass number of the ion is 16 + 16 = 32. Because the ion has 16 protons and 18 electrons, its net charge is 2–.Why does oxygen have 2 lone pairs?
This method works because each covalent bond that an atom forms adds another electron to an atoms valence shell without changing its charge. For example, hydrogen typically makes one bond because its full valence shell is 2 and its valence number is 1. Oxygen typically has 4 non-bonding electrons (or 2 lone pairs).Why does sulfate have 4 Oxygens?
Sulfate is a compound containing a sulfur atom surrounded by four oxygen atoms. The sulfate compound forms a tetrahedral arrangement with bonding. Sulfate forms a salt when converted to sulfuric acid. Two double bonds and two single bonds are used to link oxygen to the sulfur atom.How do you find the number of protons in an ion?
A positively-charged ion or cation has more protons than electrons. The proton number is the atomic number of the element, while the electron number is the atomic number minus the charge. A negatively-charged ion or anion has more electrons than protons. Again, the number of protons is the atomic number.What types of electron pairs would you find surrounding the central atom of the sulfate ion?
Answer : The type of electron pair present in the surrounding of the central atom of the sulfate ion is, bonding electron pair. Explanation : Lewis-dot structure : It shows the bonding between the atoms of a molecule and it also shows the unpaired electrons present in the molecule.Which of the following atoms would prefer to form three bonds and have one lone pair?
Fluorine (and all halogens) tends to form one bond and have 3 lone pairs. With these electron configurations, none of these atoms will have any formal charge.Why Valency of sulphate is 2?
Because there are three sulfate ions in the formula, 6 divided by 3 yields a valence number of 2 for sulfate. Aluminum makes ions with a positive charge, which is why the sulfate ion has a negative charge, and that makes the sulfate radical have a 2-valency.Why is so4 negatively charged?
Now since the two H+ ions leave 2 electrons behind the (SO)4 gets these two electrons and hence acquires two negative charge. It has -2 sign due to formal negative charge on oxygen. Formal charge of an atom = number of it's valence electron before bonding - number of it's valence electron after bonding.What is the bond order of so42?
The average bond order in the sulfate ion is 1.5.What is the Valency of sulphate ion?
For example, H2SO4 has two hydrogen atoms, so the valency of sulfate is two. Because sulfate can bond with two positive hydrogen atom, its valency is the opposite charge and often expressed as 2-.