How many oxygens are in a sulfide ion?
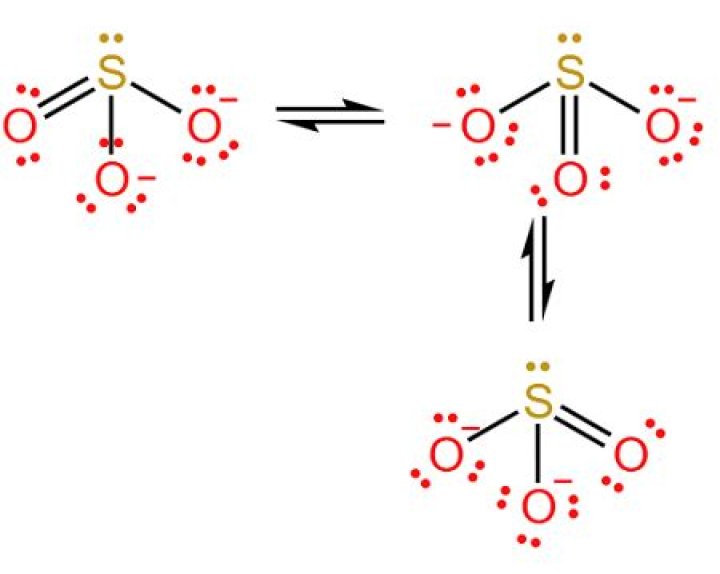
.
Also know, what is sulfide ion?
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Sulfide also refers to chemical compounds large families of inorganic and organic compounds, e.g. lead sulfide and dimethyl sulfide.
Beside above, how many bonds does oxygen form? two
Consequently, how many electrons does a sulfide ion have?
In your case, the sulfide anion, S2− , carries a (2−) negative charge, which can only mean that it gained electrons. More specifically, it gained 2 electrons. A neutral sulfur atom has an atomic number equal to 16 , which means that it has 16 protons inside its nucleus and 16 electrons surrounding its nucleus.
Does sulfide have oxygen?
Sulfide. Sulfide is the most reduced form of the sulfur compounds and, despite the fact that sulfide can be chemically oxidized in the presence of oxygen, it can also be spontaneously oxidized under anaerobic conditions using an abiotic anode as electron acceptor.
Related Question AnswersWhat is the symbol for sulfur ion?
symbol SIs sulfide an ion?
Inorganic sulfides are ionic compounds containing the negatively charged sulfide ion, S2−; these compounds may be regarded as salts of the very weak acid hydrogen sulfide. Organic sulfides are compounds in which a sulfur atom is covalently bonded to two organic groups.Where is h2s commonly found?
Hydrogen Sulfide (H2S) is a gas commonly found during the drilling and production of crude oil and natural gas, plus in wastewater treatment and utility facilities and sewers. The gas is produced as a result of the microbial breakdown of organic materials in the absence of oxygen.What is sulfide used for?
Hydrogen sulfide is used primarily to produce sulfuric acid and sulfur. It is also used to create a variety of inorganic sulfides used to create pesticides, leather, dyes, and pharmaceuticals. Hydrogen sulfide is used to produce heavy water for nuclear power plants (like CANDU reactors specifically).Is h2s an organic compound?
In H2S , it is hydrogen sulfide, and no carbon is present, and so it cannot be an organic compound. That leaves hydrogen sulfide gas as an inorganic compound.What is the name of HS?
Bisulfide (systematically named sulfanide and hydrogen(sulfide)(1−)) is an inorganic anion with the chemical formula HS− (also written as SH−). It contributes no color to bisulfide salts, and its salts may have a distinctive putrid smell.Is Sulphur an anion or cation?
Halogens always form anions, alkali metals and alkaline earth metals always form cations. Most other metals form cations (e.g. iron, silver, nickel), whilst most other nonmetals typically form anions (e.g. oxygen, carbon, sulfur).What is the difference between sulfide and sulphide?
Both are sulphide (British English) and sulfide (American English) are both perfectly acceptable. If this continues then sulfide may become the accepted British English spelling too. In any case language is fluid and irrelevant to the chemistry.Is sulfur a positive or negative ion?
Sulfur is in group 6 of the periodic table. What is the charge on its ions, and is the charge positive or negative? The charge is negative, since sulfur is a non-metal. The charge on the ion is (8 - 6) = 2.Is Lithium a cation or anion?
As an element, lithium is uncharged, so it is neutral, and neither a cation nor an anion. In aqueous solution, lithium is an Li+ cation, as is typical of Group I elements such as Li, Na, KWhy is oxygen negative 2?
Because electrons have a charge of 1-, adding two electrons would make the charge of the oxide ion 2-. Oxygen takes the electron density from other atoms when it forms two bonds, to become a negative two ion.Will K form a negative ion?
It would tend to lose two electrons and form a +2 ion. Potassium is in the first column and therefore has 1 electron in its outermost shell. It would tend to lose one electron and form a +1 ion.How many neutrons are in magnesium?
| Name | Magnesium |
|---|---|
| Atomic Mass | 24.305 atomic mass units |
| Number of Protons | 12 |
| Number of Neutrons | 12 |
| Number of Electrons | 12 |