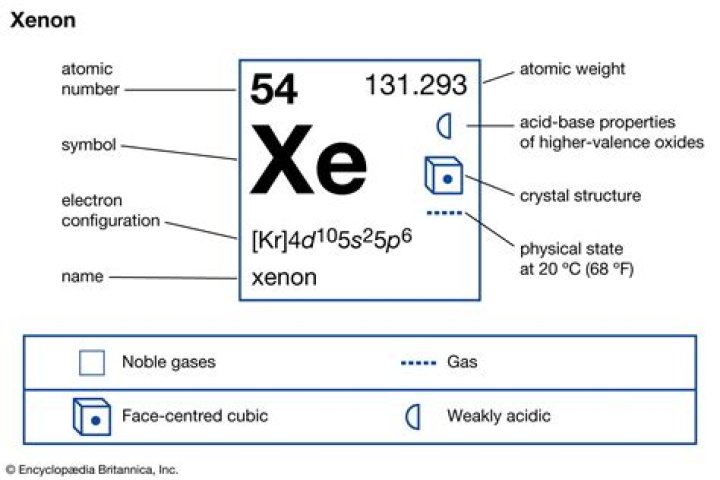
| Name | Xenon |
| Atomic Mass | 131.29 atomic mass units |
| Number of Protons | 54 |
| Number of Neutrons | 77 |
| Number of Electrons | 54 |
.
Also know, how many isotopes does xenon have?
Naturally occurring xenon (54Xe) consists of seven stable isotopes and two very long-lived isotopes.
Beside above, how many protons and neutrons does xenon have? 54
Also Know, what element has a mass number of 131?
Iodine I-131
How many protons neutrons and electrons does XE 135 possess?
Xenon-135
| General |
| Protons | 54 |
| Neutrons | 81 |
| Nuclide data |
| Natural abundance | syn |
Related Question Answers
Why is Xenon so expensive?
Why are they so expensive? Xenon HID bulbs are more expensive than halogens due to the different technology and gases used to produce the bulb but on the flip side they do last a lot longer than halogen bulbs.Is Xenon heavier than air?
Xenon (Xe), chemical element, a heavy and extremely rare gas of Group 18 (noble gases) of the periodic table. More than 4.5 times heavier than air, xenon is colourless, odourless, and tasteless.Is Xenon poisonous?
Xenon is not considered to be toxic but many of its compounds are toxic as a result of their strong oxidizing properties. Characteristics: Xenon is a rare, colorless, odorless heavy gas. Many compounds of xenon have now been made, principally with fluorine or oxygen.Is xenon magnetic?
Xenon is a chemical element with the symbol
Xe and atomic number 54. It is a colorless, dense, odorless noble gas found in Earth's atmosphere in trace amounts.
| Xenon |
| Magnetic ordering | diamagnetic |
| Magnetic susceptibility | −43.9·10−6 cm3/mol (298 K) |
| CAS Number | 7440-63-3 |
| History |
Why Xenon is called Stranger gas?
Travers discovered this element in 1898. The element's name came from the Greek word xenos, which means 'stranger'. Xenon belongs to the group of the noble gases. The gas is not very reactive, because if fulfills the octet rule.Is Xenon 131 stable?
Xenon is a heavy rare gas with nine stable isotopes, 124Xe, 126Xe, 128Xe, 129Xe, 130Xe, 131Xe, 132Xe, 134Xe and 136Xe, five of which are radiogenic. Xe is produced by beta decay of 129I (half-life = 16 Myr); 131Xe, 132Xe, 134Xe and 136Xe are fission products of both 238U and the extinct nuclide 244Pu.Why is Xenon so unreactive?
Chemists originally believed that the noble gases could not form compounds , because their full valence shell of electrons made them chemically stable and unreactive. The inner electrons shield the outermost electrons, so they are less strongly attracted to the nucleus.Who found xenon?
Morris Travers William RamsayCan you tell from the periodic table exactly how many neutrons are in an atom?
Because we can find the number of protons and the atomic mass of an atom by looking at its element information in the periodic table, we can calculate the number of neutrons in that atom by subtracting the number of protons from the atomic mass.What is atomic mass number?
The mass number (symbol A, from the German word Atomgewicht [atomic weight]), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The mass number is different for each different isotope of a chemical element.What is the charge of a proton?
Proton Charge While a proton has a charge of +1, or 1e, an electron has a charge of -1, or -e, and a neutron has no charge, or 0e. 1 elementary charge is equal to: 1.602 x 10^-19 coulombs.What element has 16 neutrons?
phosphorus
How many neutrons does gold have?
118 neutrons
Is Xenon a neutral atom?
Xenon atoms have 54 electrons and the shell structure is 2.8. The ground state electronic configuration of neutral xenon is [Kr].What does xenon look like?
Its atomic number is 54 and it is a member of the noble gases, which are found in Group 18 on the periodic table. Xenon is odorless, colorless, and heavier than the air found in our atmosphere. Because of its ability to emit light when electricity is added, it can be used in specialized lamps.What type of element is oxygen?
Oxygen is the eighth element of the periodic table and can be found in the second row (period). Alone, oxygen is a colorless and odorless molecule that is a gas at room temperature. Oxygen molecules are not the only form of oxygen in the atmosphere; you will also find oxygen as ozone (O3) and carbon dioxide (CO2).What is xenon atomic number?
54
How many neutrons are in CS?
Diagram of the nuclear composition and electron configuration of an atom of caesium-133 (atomic number: 55), the most common isotope of this element. The nucleus consists of 55 protons (red) and 78 neutrons (blue).