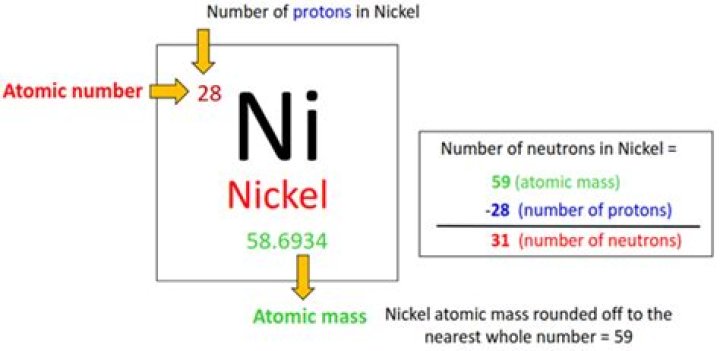
Nickel is the first element in the tenth column of the periodic table. It is classified as a transition metal. Nickel atoms have 28 electrons and 28 protons with 30 neutrons in the most abundant isotope..
Hereof, what element contains 15 electrons and 19 neutrons?
Phosphorus
Furthermore, is nickel a neutral atom? Nickel is an element, a substance that consists of only one type of atom, located in Group 10 of the periodic table of the elements. The number of positively charged protons in nickel's nucleus means that neutral nickel atoms have 28 total electrons.
Also asked, what element has 28 electrons when neutral?
nickel
How many neutrons does nickel 58 have?
. Ni-58 has an atomic number of 28 and a mass number of 58. Therefore, Ni-58 will have 28 protons, 28 electrons, and 58-28, or 30, neutrons.In Ni-60 2+ species, the number of protons is same as in neutral Ni-58.
Related Question Answers
What is the number of valence electrons in phosphorus?
five
Which element has 15 neutrons?
Phosphorus
What element has 16 neutrons?
phosphorus
How many neutrons does nitrogen have?
7
What element has 22 protons and 22 neutrons?
A neutral atom of argon also has 22 Neutrons.What is the number of neutrons in phosphorus?
16 neutrons
How do you get the number of neutrons?
This means to find the number of neutrons you subtract the number of protons from the mass number. On the periodic table, the atomic number is the number of protons, and the atomic mass is the mass number.What element contains 28 protons and 30 neutrons?
Nickel is the first element in the tenth column of the periodic table. It is classified as a transition metal. Nickel atoms have 28 electrons and 28 protons with 30 neutrons in the most abundant isotope.How do you find the electrons in an element?
The number of electrons in a neutral atom is equal to the number of protons. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. The number of neutrons is equal to the difference between the mass number of the atom (M) and the atomic number (Z).How do you find the amount of electrons in an isotope?
Understand that isotopes of an element have different mass numbers but the same number of protons. Using the Periodic Table, find the atomic number of the element. The atomic number equals the number of protons. In a balanced atom, the number of electrons equals the number of protons.Can you tell from the periodic table exactly how many neutrons are in an atom?
Because we can find the number of protons and the atomic mass of an atom by looking at its element information in the periodic table, we can calculate the number of neutrons in that atom by subtracting the number of protons from the atomic mass.How many electrons are in an element?
Atoms must have equal numbers of protons and electrons. In our example, an atom of krypton must contain 36 electrons since it contains 36 protons. Electrons are arranged around atoms in a special way.What is nickels freezing point?
1,455 °C
Why does nickel have 10 valence electrons?
Nickel has eight electrons in the 3d orbital and two electrons in the 4s orbital, which means nickel has 10 total valence electrons. The reason it has 10 is because nickel is a transition metal, so the d and s electrons can participate in chemical bonding.What are the outermost electrons in nickel?
The valence shell is the outermost shell of an atom in its uncombined state, which contains the electrons most likely to account for the nature of any reactions involving the atom and of the bonding interactions it has with other atoms. Therefore Ni has 2 valence electrons.What is nickels density?
Atomic Number: 28. Atomic Weight: 58.6934. Melting Point: 1728 K (1455°C or 2651°F) Boiling Point: 3186 K (2913°C or 5275°F) Density: 8.912 grams per cubic centimeter.Why is nickel ductile?
Nickel is very strong and resistant to corrosion, making it excellent for strengthening metal alloys. It is also very ductile and malleable, properties that allow its many alloys to be shaped into wire, rods, tubes, and sheets.Why is nickel named after the Devil?
Nickel is the only element named after the devil. The name comes from the German word Kupfernickel, meaning "Old Nick's copper," a term used by German miners. They tried to remove copper from an ore that looked like copper ore, but they were unsuccessful. Nickel is also used in the manufacture of many other alloys.