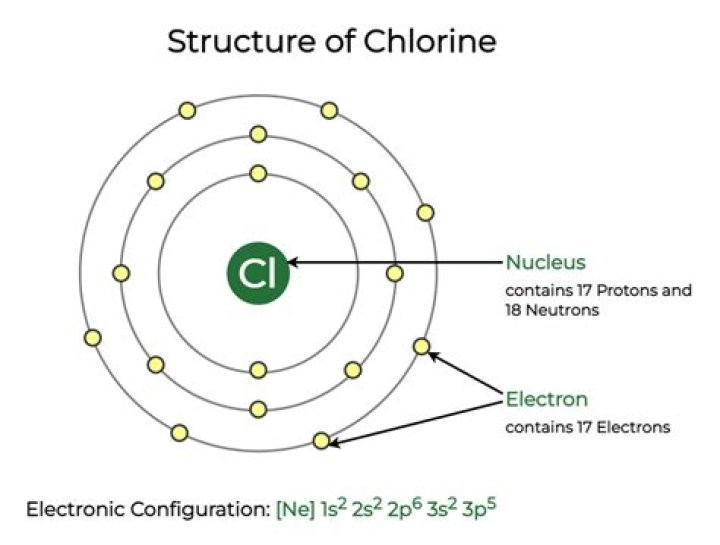
18 neutrons
.
Likewise, how do you find the number of neutrons in chlorine?
Each proton and neutron has a relative mass of 1 unit. Therefore we can subtract the number of protons from the atomic mass number in order to work out the number of neutrons. Chlorine has a mass number of 35, minus the 17 protons means that chlorine has 18 neutrons.
Subsequently, question is, how many electrons does chlorine have? 17 electrons
One may also ask, how many neutrons does chlorine atom have?
17
Do most chlorine atoms have 18 neutrons or 20 neutrons?
Not all atoms of an element, therefore, have the same atomic mass number. All chlorine atoms, for instance, have 17 protons. However, some chlorine atoms have 18 neutrons, while other chlorine atoms have 20 neutrons. Atoms of chlorine with 18 and 20 neutrons are called chlorine isotopes.
Related Question Answers
How many neutrons are in magnesium?
There are 13 neutrons in an atom of magnesium-25. We can determine this by subtracting the number of protons in the atom from the atomic mass, whichHow many neutrons does nitrogen have?
7
What are the 2 isotopes of chlorine?
The element chlorine has two isotopes, chlorine–35 and chlorine–37. The abundance of these isotopes when they occur naturally is ( ext{75}\%) chlorine–35 and ( ext{25}\%) chlorine–37. Calculate the average relative atomic mass for chlorine.How many protons neutrons and electrons are in chlorine 35?
35-Chlorine has 17 Protons, and 18 Neutrons in its Nucleus. 37-Chlorine has 17 Protons, and 20 Neutrons in its Nucleus. Both, of these main & stable isotopes have a total of 17 Electrons.How do you get the number of neutrons?
This means to find the number of neutrons you subtract the number of protons from the mass number. On the periodic table, the atomic number is the number of protons, and the atomic mass is the mass number.How much does pure chlorine cost?
Powder Chlorine (Calcium Hypochlorite) - contains a maximum of 70% available chlorine. Strength is rapidly lost in storage. Average cost per pound of powder chlorine is $1.23‡. Therefore, the cost per pound of AVAILABLE CHLORINE = $1.76/lb.Is the symbol for chlorine Cl or CI?
Chlorine is a chemical element with the symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine is a yellow-green gas at room temperature.How many electrons are in a neutral atom of chlorine 37?
17 electrons
How many neutrons does chlorine 35?
18 neutrons
What is the cost of sodium?
Metallic sodium is priced at about 15 to 20 cents/lb in quantity.Can you tell from the periodic table exactly how many neutrons are in an atom?
Because we can find the number of protons and the atomic mass of an atom by looking at its element information in the periodic table, we can calculate the number of neutrons in that atom by subtracting the number of protons from the atomic mass.Why is cl 35 more abundant?
In other words, in every 100 chlorine atoms, 75 atoms have a mass number of 35, and 25 atoms have a mass number of 37. This is because the chlorine-35 isotope is much more abundant than the chlorine-37 isotope. The table shows the mass numbers and abundances of naturally-occurring copper isotopes.How many neutrons does s 36 have?
16
How many neutrons does oxygen have?
8
What is chlorine used for?
Chlorine is commonly used as an antiseptic and is used to make drinking water safe and to treat swimming pools. Large amounts of chlorine are used in many industrial processes, such as in the production of paper products, plastics, dyes, textiles, medicines, antiseptics, insecticides, solvents and paints.What is the number of protons in chlorine?
17
Is sodium a metal?
Sodium is an element that is a member of the alkali metal group with a symbol Na. It is physically silver colored and is a soft metal of low density. Pure sodium is not found naturally on earth because it is a highly reactive metal.What is the number of valence electrons in chlorine?
7 valence electrons
What is the Valency of chlorine?
The electronic configuration of chlorine can be written as 2, 8, 7. 2, 8, 7 electrons are distributed in the shells K, L, M respectively. Therefore, valence electron in chlorine is 7 and it needs to gain 1 electron from the outermost orbit to attain octet. Hence, the valency of chlorine is 1.