news /
How many energy levels does antimony have?
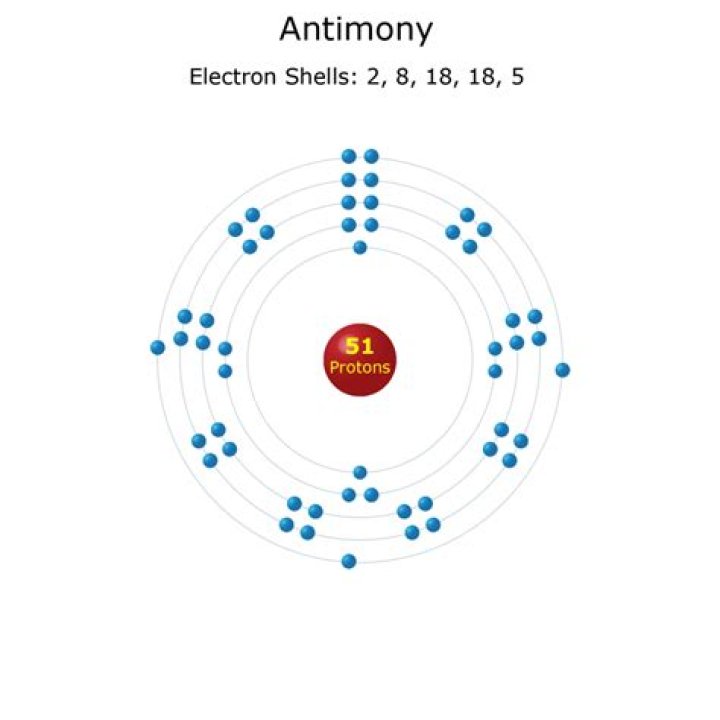
Data Zone
| Classification: | Antimony is a metalloid |
|---|---|
| Electrons: | 51 |
| Protons: | 51 |
| Neutrons in most abundant isotope: | 70 |
| Electron shells: | 2,8,18,18,5 |
.
Correspondingly, what is the energy level of antimony?
| Number of Energy Levels: | 5 |
|---|---|
| First Energy Level: | 2 |
| Second Energy Level: | 8 |
| Third Energy Level: | 18 |
| Fourth Energy Level | 18 |
Additionally, how many protons neutrons and electrons are in the antimony III ion? The nucleus consists of 51 protons (red) and 70 neutrons (yellowe). 51 electrons (white) bind to the nucleus, successively occupying available electron shells (rings).
Additionally, which elements have the same number of energy levels?
For example, elements in the same group have the same number of valence electrons or valency. Elements in the same period or rows have similar number of energy levels. For example, potassium and bromine belong to the same period in the periodic table and thus have the same number of energy levels.
What is antimony found in?
3000 BC
Related Question AnswersIs there an element 124?
Unbiquadium, also known as element 124 or eka-uranium, is the hypothetical chemical element with atomic number 124 and placeholder symbol Ubq. Unbiquadium and Ubq are the temporary IUPAC name and symbol, respectively, until the element is discovered, confirmed, and a permanent name is decided upon.What is the second most reactive metal?
So we just covered the alkali metals in Group I. You will find the alkaline earth metals right next door in Group II. This is the second most reactive family of elements in the periodic table.What is the ionization energy of SB?
First Ionization Energy of Antimony is 8.64 eV.Is antimony a poison?
Antimony toxicity occurs either due to occupational exposure or during therapy. Occupational exposure may cause respiratory irritation, pneumoconiosis, antimony spots on the skin and gastrointestinal symptoms. In addition antimony trioxide is possibly carcinogenic to humans.What element has 51 neutrons?
AntimonyIs SB a neutral atom?
Antimony: properties of free atoms. Antimony atoms have 51 electrons and the shell structure is 2.8. The ground state electron configuration of ground state gaseous neutral antimony is [Kr].What is unique about Antimony?
Antimony (Sb) has an atomic number of fifty-one, with fifty-one protons in the nucleus. It is a highly brittle, bluish-white, semi-metallic element. Interesting Antimony Facts: Antimony was used in ancient Egypt as a form of eyeliner (kohl).What is the valence of selenium?
Selenium specifically has an electron configuration of 2-8-18-6. The six electrons in the outermost shell allow selenium to have a variety of valence numbers. Selenium compounds have been found that have valences of -2, 4, and 6. Speaking of the number six, selenium is found to have six naturally occurring isotopes.Which element has the highest first ionization energy?
From this trend, Cesium is said to have the lowest ionization energy and Fluorine is said to have the highest ionization energy (with the exception of Helium and Neon).What is another word for energy level?
Definition of energy level. : one of the stable states of constant energy that may be assumed by a physical system —used especially of the quantum states of electrons in atoms and of nuclei. — called also energy state.How are energy levels calculated?
Number of energy levels in each period- The atoms in the first period have electrons in 1 energy level.
- The atoms in the second period have electrons in 2 energy levels.
- The atoms in the third period have electrons in 3 energy levels.
- The atoms in the fourth period have electrons in 4 energy levels.
Which element has the highest energy level?
Electrons| Atomic Number | Element | Energy Levels or "shells" |
|---|---|---|
| N | ||
| 1 | Hydrogen (H) | |
| 2 | Helium (He) | |
| 3 | Lithium (Li) |



