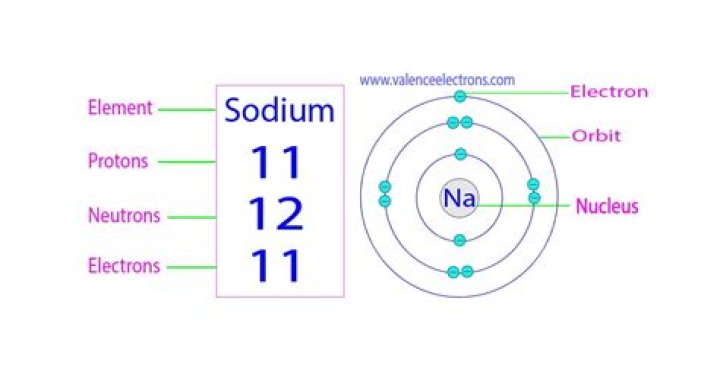
When atoms lose or gain electrons they arecalled . If an atom gains an it is called an anion. When an atomloses an electron it is called a . A sodium atom haseleven and a atom has seventeen electrons..
Just so, how many electrons does sodium gain or lose?
Cations. A neutral sodium atom is likely toachieve an octet in its outermost shell by losing its onevalence electron. The cation produced in this way,Na+, is called the sodium ion to distinguish itfrom the element. The outermost shell of the sodium ion isthe second electron shell, which has eight electronsin it.
Also, which groups lose or gain electrons? The alkali earth metals (group 2), such as, Mg orSr lose two e-'s to attain the configuration of Ne. Thus,the group 2 metals tend to have a +2 charge. On the otherside of the periodic table elements gain electrons toresemble the next higher noble gas.
Likewise, does NA gain or lose electrons?
A cation is an atom that has lost a valenceelectron and therefore has more positive protons thannegative electrons, so it is positively charged. An anion isan atom that has gained a valence electron and is negativelycharged. Sodium will easily lose that extraelectron. When it does, it becomesunbalanced.
What is sodium called when it loses an electron?
The Sodium ion. In the reaction the Sodium atom loses anelectron to the Chlorine atom. Thus it has one more proton thanit has electron and has a net positive charge of +1 and itis now called the Sodium ion.
Related Question Answers
What is the weakest bond?
The ionic bond is generally the weakest ofthe true chemical bonds that bind atoms toatoms.How many electrons does calcium gain or lose?
2 electrons
What is Sodiums charge?
Sodium atoms have no charge, sodiumhas eleven protons in its nucleus and eleven electrons orbiting it.This makes it electrically neutral. When sodium forms anion, it will form Na+ a cation. This is because sodium hasone valence electron (one electron in its outershell).Why carbon do not lose or gain 4 electrons?
Carbon cannot gain or loseelectrons. Carbon cannot for C4-because If itgains 4 electrons, it would be difficult for thenucleus with six protons to hold on to tenelectrons, that is, four extraelectrons.Why does sodium want to lose an electron?
The sodium atom and the chlorine atom wantto make their outside shells contain only eight electrons.The sodium atom wants to lose an electron and thechlorine atom wants to gain an electron. When the twoatoms come together the electron from the sodium atomjumps into the gap in the outer shell of the chlorineatom.Why does taking away electrons increase the charge?
However, atoms can gain or lose electrons:increasing or decreasing the negative charge.Ionisation is the addition or removal of an electronto create an ion. An atom's charge can only change throughgaining or losing electrons. Protons cannot be added orremoved because they are bound up with neutrons in thenucleus.What is the most stable electron configuration?
The most stable electron configuration is that ofa noble gas, due to the fact that its valence shell is filled. Forhelium, that means two valence electrons (a duet) in the 1ssublevel, and for the rest it means eight valence electrons(an octet) in the outermost s and p sublevels.What is meant by covalent bond?
A covalent bond, also called a molecularbond, is a chemical bond that involves the sharing ofelectron pairs between atoms. These electron pairs are known asshared pairs or bonding pairs, and the stable balance ofattractive and repulsive forces between atoms, when they shareelectrons, is known as covalent bonding.Which element would be most likely to gain electrons?
Nonmetals (the right 1/3 of the Chart) when they formionic compounds do so by gaining electrons. This due to thefact that they have high ionization energies and highelectron affinities (referring to their ability to collectelectrons). When they do form ions, they are of negativecharge and are called anions.What happens when an atom loses all of its electrons?
However, if something happens to make an atomlose or gain an electron then the atom will nolonger be neutral. An atom that gains or losesan electron becomes an ion. If it gains a negativeelectron, it becomes a negative ion. If it loses anelectron it becomes a positive ion (see page 10 for more onions).How many electrons does the neutral atom gain or lose?
How many electrons does the neutral atom gain orlose when each ion forms? 4.How do electrons get their charge?
Charge It! Electrons are the negatively chargedparticles of atom. Together, all of the electrons of an atomcreate a negative charge that balances the positivecharge of the protons in the atomic nucleus.Electrons are extremely small compared to all of the otherparts of the atom.Which family of elements loses electrons most easily?
In particular, cesium (Cs) can give up its valenceelectron more easily than can lithium (Li). In fact,for the alkali metals (the elements in Group 1), the ease ofgiving up an electron varies as follows: Cs > Rb > K> Na > Li with Cs the most likely, and Li the leastlikely, to lose an electron.How many valence electrons are in each group?
The Group 1 atoms have 1 valence electron.The Group 2 atoms have 2 valence electrons. TheGroup 3 atoms have 3 valence electrons. TheGroup 4 atoms have 4 valence electrons.How many electrons does magnesium gain or lose?
two electrons
Would nitrogen expect to gain electrons?
With two electrons it keeps for itself and sixthat it shares with another atom, that makes eight electronstotal. Thus this form of nitrogen is more stable than asingle atom of nitrogen, which only has five valenceelectrons and must react with another chemical to becomestable.How do you tell if an element gains or loses electrons?
Just determine whether it is easier tolose the electrons to become stable or gainthem. If they have less than three valence electrons,it is easier to lose them and become a positively chargedion. If they have more than four valence electrons,it is easier to gain electrons and become negativelycharged.Is it easier to lose or gain electrons?
Low ionization energy means that it easy to loseelectrons. The elements on the left side lose theirelectrons fairly easily and the elements on the right sideof the periodic table do not lose their electronsvery easily. The ability to gain electrons is also relatedto the position on the periodic table.