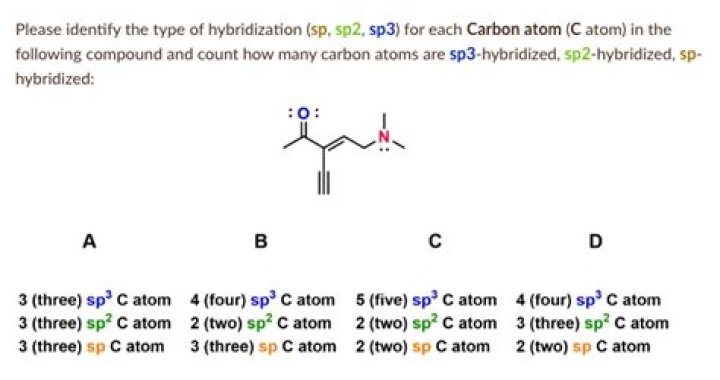
Answer and Explanation: The carbon atom, that forms four sigma bonds, is sp3 s p 3 hybridized carbon. In the given compound there are five carbon atoms that form four.
Moreover, how many atoms are sp3 hybridized?
A carbon bonded to 4 different atoms is considered to be sp3 hybridized.
how many carbon atoms in menadione are sp2 hybridized? That makes 24 sp2 hybridized carbon atoms.
Similarly, what are sp3 hybridized carbons?
In the excited carbon atom its one s-orbital (2s) and three p-orbitals (2px , 2py, 2pz) intermix and reshuffle into four identical orbitals known as sp3 orbitals. Thus, carbon atom undergoes sp3 hybridisation. (because they are made from one 's-orbital' and three 'p-orbitals' they are called sp3 orbitals).
What is the bond order of CO?
The bond order of CO is 3. Bond order is the number of chemical bonds between a pair of atoms . For example, in diatomic nitrogen N≡N the bond order is 3, in acetylene H−C≡C−H the bond order between the two carbon atoms is also 3, and the C−H bond order is 1.
Related Question Answers
Is h2o polar or nonpolar?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.What is the bond order of f2?
The bond order is 1/2(no. of bonding electrons - no. of antibonding So in order of stability you have; F2+,F2.So,F2+>F2.Is water sp2 or sp3?
of a hydrogen atom on methane. They may also contain lone pairs—this explains the geometry of water, which is sp3 hybridized due to the lone pair, which occupies one of the four hybrid orbitals. Again, the bond angle is slightly distorted due to the electron repulsion of this lone pair.Is f2 diamagnetic or paramagnetic?
It's paramagnetic because it posses 2 unpaired electrons. For Difluorine, by counting the number bonding, 10, and number of antibonding, 8, give us the BO of 1. It is diamagnetic with no unpaired electrons.What is SP sp2 sp3?
sp hybridization occurs due to the mixing of one s and one p atomic orbital, sp2 hybridization is the mixing of one s and two p atomic orbitals and sp3 hybridization is the mixing of one s and three p atomic orbitals.Does hybridization include lone pairs?
Lone pairs are electron groups which counts towards hybridization. Lone pairs count as one electron group towards total hybridization. Oxygen has two lone pairs. Along with the two bonded atoms, the hydrogen's, the central atom has a total of four electron groups, giving the central atom an sp3 hybridization.Is sp3 oxygen hybridized?
The oxygen is sp3 hybridized which means that it has four sp3 hybrid orbitals. One of the sp3 hybridized orbitals overlap with s orbitals from a hydrogen to form the O-H signma bonds. Due to the sp3 hybridization the oxygen has a tetrahedral geometry.Is ch2 sp3 hybridized?
No, the CH2 planes are mutually perpendicular to each other. The center C atom is sp hybridized and is involved in two π bonds. The p orbitals used to form each π bond must be perpendicular to each other.Why is oxygen sp3 hybridized?
The oxygen is sp3 hybridized which means that it has four sp3 hybrid orbitals. One of the sp3 hybridized orbitals overlap with s orbitals from a hydrogen to form the O-H signma bonds. Due to the sp3 hybridization the oxygen has a tetrahedral geometry.Is ammonia a sp2 or sp3?
Ammonia ( NH3) , or, more accurately, the central atom in ammonia, is sp3 hybridized. Here's how you'd go about determining this. First, start with NH3 's Lewis structure, which must account for 8 valence electrons - 5 from nitrogen and 1 from each hydrogen atom.What does it mean to be sp3 hybridized?
The term “sp3 hybridization” refers to the mixing character of one 2s-orbital and three 2p-orbitals to create four hybrid orbitals with similar characteristics. In order for an atom to be sp3 hybridized, it must have an s orbital and three p orbitals.Are single bonds sp3?
Basically sp3 , sp2 , sp are hybridization states …it is a process of forming hybrid orbitals . Which means Carbon has used its four valence electrons to form single bond , means all the bonds are same so we will call it sp3 as one s and 3 p orbitals sum up to four and you are having four bonds.What does sp2 mean?
SP2 hybridization means the overlap of 1 S orbital and 2 P orbitals to form three orbitals, each have the name SP2, the new hybridized orbitals are stronger than the original ones, so they are capable of forming a strong bond. Exampes on SP2 hybridiztaion: Ethylene molecule ( C2H4)How sp3 hybridization takes place?
Now a sp3 hybridisation takes place when one s-orbital and 3-p orbitals are mixed together, thereby resulting in four sp3 hybridised orbitals. This energy can either be externally provided or it can be accumulated during collisions among molecules/atoms. In hybridization, the electrons of carbon are excited.Are Carbanions sp2?
A carbanion is formed by removing one group from a tetrahedral carbon, but leaving the bonding pair with the carbon. If you recall from your VSEPR theory, central atoms with three electron pairs, all bonding, have a trigonal planar geometry. Trigonal planar is a characteristic of sp2 hybridization.How do you determine polarity?
(If the difference in electronegativity for the atoms in a bond is greater than 0.4, we consider the bond polar. If the difference in electronegativity is less than 0.4, the bond is essentially nonpolar.) If there are no polar bonds, the molecule is nonpolar.What is the hybridization of benzene?
Carbon atoms in the benzene ring have a trigonal planar geometry around them since the carry bonds with three other groups and therefore, the hybridization is sp2 . This hybridization is a must to achieve the bond angle 120∘ which is found in benzene rings.Does oxygen hybridize?
Answer: The oxygen atom must have either sp2 or sp hybridization, because it needs a p orbital to participate in the C–O π bond. This oxygen atom has three attachments (the carbon and two lone pairs), so we use sp2 hybridization.How many sp2 hybridized carbon atoms are in benzene?
three sp
2 hybridized