How long does salt water battery last?
.
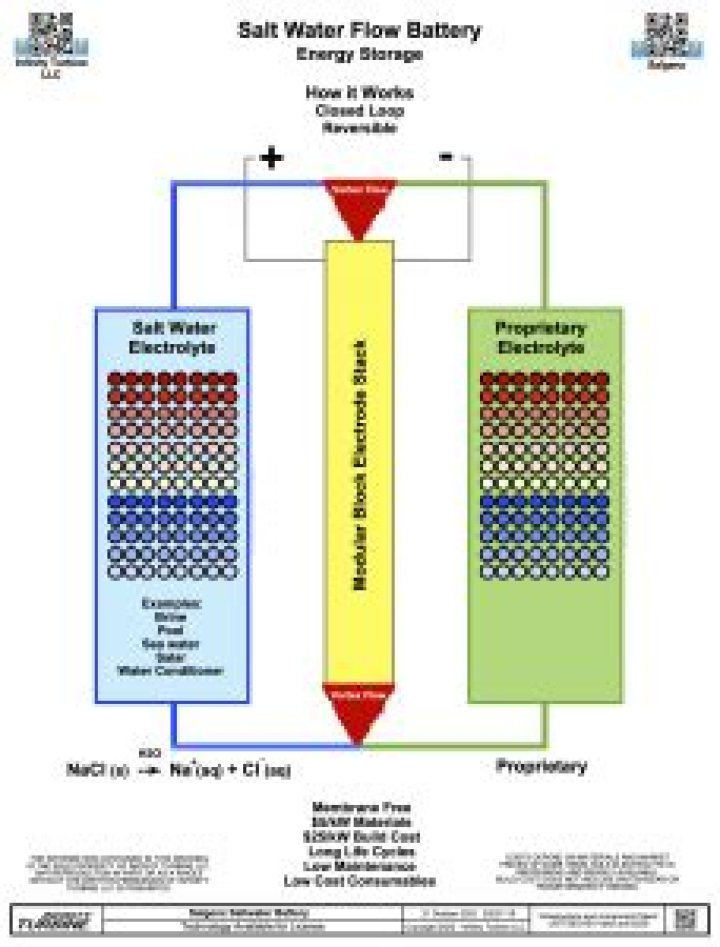
Similarly, you may ask, how do salt water batteries work?
A salt water battery is a wet-cell battery that uses a reaction with salt water, air, and a magnesium anode to produce electricity. The unique characteristic of the salt water battery is that it uses the air as a cathode, so there is no need for separate half-cells for each electrode as in other wet-cell batteries.
Additionally, why must salt be added to the water for the battery to work? When salt water flows over the electrodes, they capture the ions, producing a charged battery. When the salt water is replaced by freshwater, the cycle can be reversed, but in order to do so, electrons have to flow between the two electrodes, creating a usable current.
Hereof, how do you make a salt water battery stronger?
Prepare the saltwater electrolyte for your zinc-air battery.
- Place the bowl on your scale and put the balance back to zero (tare the scale).
- Weigh 25 grams (g) of table salt (NaCl) into the bowl.
- Fill your measuring cup with 500 milliliters (mL) of tap water.
- Add the water into the bowl with your weighed salt.
Are salt water batteries rechargeable?
Inexpensive and stable—The salt water battery. Water could form the basis for future particularly inexpensive rechargeable batteries. Water is inexpensive, available everywhere, non-flammable and can conduct ions. However, water has one major drawback: It is chemically stable only up to a voltage of 1.23 volts.
Related Question AnswersHow can I make a homemade battery stronger?
Steps- Gather your materials.
- Fill the plastic cup roughly 3/4 full with soda.
- Make sure the soda can is completely empty.
- Cut a strip of aluminum from the soda can.
- Sand the aluminum strip (optional).
- Place the strips into the solution.
- Attach lead wires to metal strips.
- Test the battery.
Can we generate electricity from saltwater?
“The electrolysis method was used to produce the electricity from saltwater. Distilled water is pure and free of salts; thus it is a very poor conductor of electricity. By adding ordinary table salt to distilled water, it becomes an electrolyte solution that can conduct electricity.”Can salt water power a car?
The salt water provides the electrolyte used in a chemical reaction inside a fuel cell. This chemical reaction creates electricity, similar to how a battery creates electricity. This electricity runs a small motor which powers the car.How do you make a salt battery?
Saltwater Battery. Place one teaspoon of salt in the ceramic cup. Dispense six ounces (3/4 cup) of water into the cup and stir to dissolve the salt. Add one teaspoon of vinegar and 1/4 teaspoon of bleach into the solution; stir.How many volts does salt water produce?
One gram of water dissolves seven grams of sodium FSI. This produces a clear saline solution with an electrochemical stability of up to 2.6 volts – twice as much as other aqueous electrolytes.Can salt water charge a phone?
This is a portable charging pack that charges your phone with salt and water. The salt and water mixture will release hydrogen which can be generated into electricity, therefore making it possible to charge your phone.Is sea water an electrolyte?
Since the major components of sea water are constant throughout the oceans (The Marcet Principle), it is possible to treat ocean waters as an electrolyte solution (sea salt) with a dash of the non-electrolyte boric acid. This simplifies the physical chemistry of sea water solutions and other natural waters.How do you add water to a battery?
Use only distilled water to fill the cells. Distilled water can be purchased at most grocery stores. If the electrolyte levels in the cells are low (plates are exposed), fill each cell to just cover the plates. Then use a battery charger to recharge the battery, or just drive the car for a few days in normal service.What reacts with salt water?
Word equations – The reaction between acids and alkalis When an acid reacts with an alkali, a salt and water is produced: acid + alkali → salt + water An example: hydrochloric acid + sodium hydroxide → sodium chloride + water The salt that is produced depends upon which acid and which alkali react.How do you make a battery for a science project?
A few pennies and nickels, small paper-towel squares, a vinegar-salt solution, and an aluminum strip is all you need to create a coin battery. Place the aluminum strip in the middle of your plate. You will build your battery on top. This strip will make it easier to connect the multimeter later.How does Zinc react with salt water?
Sacrificial Anodes: Zinc Protecting Parts from Salt Water. This back and forth movement of the dissolved electrolytes creates a current, as the salt water breaks apart the metal parts by making them give up its electrons to the saltwater. This process is called galvanic corrosion.How do you make a rechargeable battery?
Make Your Own Rechargeable Battery- Step 1: Waste Required to Make This Battery. For this you need :-
- Step 2: Strip Off the Cell. Take dry cell and strip it off as shown in the figure .
- Step 3: Taking Out the Carbon Rod.
- Step 4: Taking Out Magnesium Dioxide.
- Step 5: Coating.
- Step 6: Inserting Electrodes.
- Step 7: Joining Wires.
- Step 8: Making Electrolyte.



