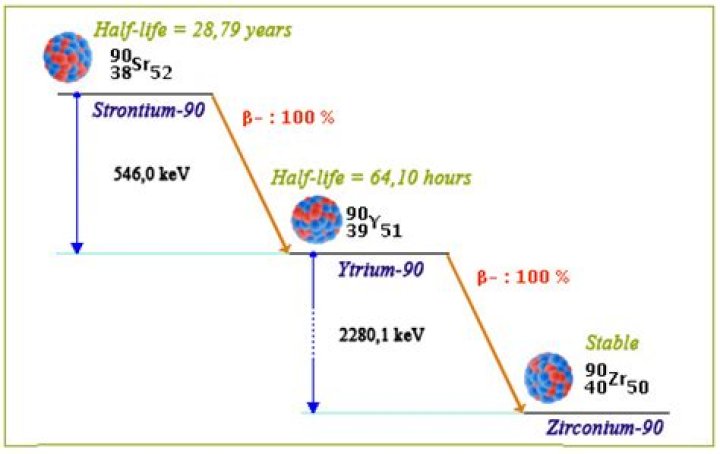
) is a radioactive isotope of strontium produced by nuclear fission, with a half-life of 28.8 years. It undergoes β− decay into yttrium-90, with a decay energy of 0.546 MeV..
Herein, how long years will it take until only 25 percent of the strontium 90 remains?
Explanation: If the half-life of Sr-90 is 25 years it means that after 25 years its amount will be half the initial quantity.
how do you calculate the half life of strontium 90? For example, the half-life of strontium-90 is 28.8 years. If you start with 10 grams of 90Sr and wait 28.8 years, you'll have 5 grams left of 90Sr. If another 28.8 years go by, 2.5 grams will remain. Another 28.8 years, and 1.25 grams will be leftand so on.
Subsequently, question is, what is the half life of strontium 90 explain your answer?
28.8 years
What is the decay rate of strontium 90?
DECAY PATHWAY: Strontium-90, half-life 28.79 years, decays via beta(-) emission (100%, 546.0 keV maximum; 195.8 keV average energy) to yttrium-90, half-life 64.00 hours; decays via beta (-) emission (99.989%, 2280.1 keV maximum, 933.7 keV average energy) to zirconium-90, half-life stable.
Related Question Answers
What is the half life for the isotope?
The rate at which a radioactive isotope decays is measured in half-life. The term half-life is defined as the time it takes for one-half of the atoms of a radioactive material to disintegrate. Half-lives for various radioisotopes can range from a few microseconds to billions of years.How many half lives have passed?
When the radioactive isotope decays to 50 % of its initial value then one half-life is said to be passed. This may vary from a few microseconds to a few billion years. Given that the rock contains 25 % of parent isotopes and 75% daughter isotopes then Let us consider the parent isotope was initially 100 %.Can you predict when an individual atom will decay?
No, its not. The decay of an individual atom is a random event. However, it is possible to predict when decay will occur based on probability, particularly when there are a lot of radioactive atoms around. Fortunately, since atoms are so small, it doesn't take much radioactive material to represent a lot of atoms.How do you calculate Half Life on a calculator?
Half-life Calculator - Exponential decay - Enter the needed values (do not use commas) Half-Life Calculation. Beginning Amount = Final Amount = Elapsed Time = Half-life =
- Final Amount Calculation. Beginning Amount = Half-life = Elapsed Time = Final Amount =
- Elapsed Time Calculation. Beginning Amount = Final Amount = Half-life = Elapsed Time =
How long does it take a 100.00 g sample of as 81 to decay to 6.25 g?
How long does it take a 100.00g sample of As-81, with a half-life of 33 seconds, to decay to 6.25g?How long is a half life for carbon 14?
Radiocarbon dating can be used on samples of bone, cloth, wood and plant fibers. The half-life of a radioactive isotope describes the amount of time that it takes half of the isotope in a sample to decay. In the case of radiocarbon dating, the half-life of carbon 14 is 5,730 years.How old is a bone if it presently contains 0.3125 of C 14?
How old is a bone if it presently contains 0.3125g of C-14, but it was estimated to have originally contained 80.000g of C-14 (half life = 5730 yr)?Why is SR 90 so dangerous?
The harmful effects of strontium-90 are caused by the high energy effects of radiation. Since radioactive strontium is taken up into bone, the bone itself and nearby soft tissues may be damaged by radiation released over time.What is the half life of cesium 137?
30.17 years
What is the half life of iodine 131?
8.02 days
What is the half life of francium 212?
Francium-216 has a half-life of 0.7 microseconds. It can emit an alpha-particle to form astatine-212 or a positron to form radon-216. Francium-212 has a half-life of 19 minutes.What does strontium 90 do to the human body?
Strontium-90 behaves like calcium in the human body and tends to deposit in bone and blood- forming tissue (bone marrow). Thus, strontium- 90 is referred to as a "bone seeker," and exposure will increase the risk for several diseases including bone cancer, cancer of the soft tissue near the bone, and leukemia.Why does strontium 90 accumulate in bones?
A large portion of the strontium will build up in bones. The harmful effects of strontium-90 are caused by the high energy effects of radiation. Since radioactive strontium is taken up into bone, the bone itself and nearby soft tissues may be damaged by radiation released over time.What is the half life of strontium 89?
50.5 days
Is strontium dangerous?
There are no harmful effects of stable strontium in humans at the levels typically found in the environment. The only chemical form of stable strontium that is very harmful by inhalation is strontium chromate, but this is because of toxic chromium and not strontium itself.Why is strontium a bone seeker?
Strontium-90 is chemically similar to calcium, and tends to deposit in bone and blood-forming tissue (bone marrow). Thus, strontium-90 is referred to as a "bone seeker." Internal exposure to Sr-90 is linked to bone cancer, cancer of the soft tissue near the bone, and leukemia.Where does cesium 137 come from?
Radioactive cesium-137 is produced spontaneously when other radioactive materials such as uranium and plutonium absorb neutrons and undergo fission. Cesium-137 is therefore a common radionuclide produced when nuclear fission, or splitting, of uranium and plutonium occurs in a reactor or atomic bomb.Who discovered strontium 90?
History of Strontium-90 Origin Non-radioactive Strontium was first detected in the mineral called Strontianite, by Adair Crawford and William Cruikshank in 1790 in Scotland. However, Sir Humphry Davy was the first person to separate metallic Strontium. Sr was discovered in the 1940s.What is strontium used for?
A soft, silvery metal that burns in air and reacts with water. Strontium is best known for the brilliant reds its salts give to fireworks and flares. It is also used in producing ferrite magnets and refining zinc. Modern 'glow-in-the-dark' paints and plastics contain strontium aluminate.