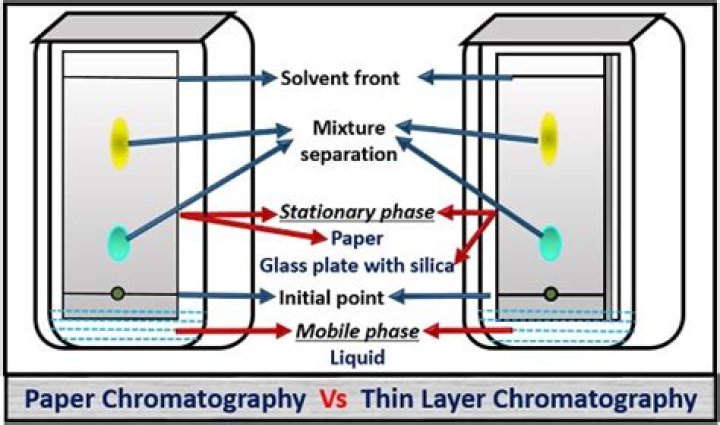
The basic difference between thin layer chromatography(TLC) and paper chromatography(PC) is that , while the stationary phase in PC is paper, the stationary phase in TLC is a thin layer of an inert substance supported on a flat, unreactive surface..
Also question is, what are the differences between thin layer chromatography and paper chromatography?
Paper chromatography requires less preparation whereas thin layer chromatography requires more preparation time. The stationary phase of paper chromatography is the water trapped in the cellulose filter paper. Thin-layer chromatography makes the use of silica gel whereas paper chromatography does not.
Furthermore, what is thin layer chromatography and how does it work? Thin-layer chromatography (TLC) is a chromatography technique used to separate non-volatile mixtures. After the sample has been applied on the plate, a solvent or solvent mixture (known as the mobile phase) is drawn up the plate via capillary action.
Beside above, what are the advantages of thin layer chromatography in comparison to paper chromatography?
The cellulose paper support in paper chromatography is flexible whereas the adsorbent in TLC is coated onto a rigid metal, glass or plastic plate. This contributes to reproducibility of spots and faster development. Due to support rigidity there is less diffusion and as a result well-defined spots are formed.
What factors affect paper chromatography?
Retention factor values in thin layer chromatography are affected by the absorbent, the solvent, the chromatography plate itself, application technique and the temperature of the solvent and plate.
Related Question Answers
What is Rf value?
The Rf value is defined as the ratio of the distance moved by the solute (i.e. the dye or pigment under test) and the distance moved by the the solvent (known as the Solvent front) along the paper, where both distances are measured from the common Origin or Application Baseline, that is the point where the sample isWhat are the uses of thin layer chromatography?
Thin layer chromatography, or TLC, is a method for analyzing mixtures by separating the compounds in the mixture. TLC can be used to help determine the number of components in a mixture, the identity of compounds, and the purity of a compound.What is the purpose of column chromatography?
Column Chromatography is a preparative technique used to purify compounds depending on their polarity or hydrophobicity. In column chromatography, a mixture of molecules is separated based on their differentials partitioning between a mobile phase and a stationary phase.Why is thin layer chromatography important?
Thin-layer chromatography (TLC) is a very commonly used technique in synthetic chemistry for identifying compounds, determining their purity and following the progress of a reaction. It also permits the optimization of the solvent system for a given separation problem.Is silica polar or nonpolar?
Silica gel, the most commonly used stationary phase, has the empirical formula SiO2. However, at the surface of the silica gel particles, the dangling oxygen atoms are bound to protons. The presence of these hydroxyl groups renders the surface of silica gel highly polar.How does polarity affect RF value?
In general, the adsorptivity of compounds increases with increased polarity (i.e. the more polar the compound then the stronger it binds to the adsorbent). Non-polar compounds move up the plate most rapidly (higher Rf value), whereas polar substances travel up the TLC plate slowly or not at all (lower Rf value).What is the principle behind paper chromatography?
Principle of paper chromatography: The principle involved is partition chromatography wherein the substances are distributed or partitioned between liquid phases. One phase is the water, which is held in the pores of the filter paper used; and other is the mobile phase which moves over the paper.What are the limitations of thin layer chromatography?
Disadvantages of TLC include application to only nonvolatile compounds, limited resolution capability (separation numbers or peak capacities of 10–50), and the absence of fully automated systems, although the individual steps of the technique can be automated.What is the stationary phase in paper chromatography?
The stationary phase in paper chromatography is the strip or piece of paper that is placed in the solvent. In thin-layer chromatography the stationary phase is the thin-layer cell. Both these kinds of chromatography use capillary action to move the solvent through the stationary phase.Why silica gel is used in TLC?
Silica gel is by far the most widely used adsorbent and remains the dominant stationary phase for TLC. The surface of silica gel with the highest concentration of geminal and associated silanols is favored most for the chromatography of basic compounds because these silanols are less acidic.What are the advantages and disadvantages of gas chromatography?
Disadvantages of gas chromatography ? Limited to volatile sample. ? Not suitable for thermally labile samples. ? Samples be soluble and don't react with the column. ? During injection of the gaseous sample proper attention is required.What are the uses of paper chromatography?
Paper chromatography is used as a qualitative analytical chemistry technique for identifying and separating colored mixtures like pigments. It is used in scientific studies to identify unknown organic and inorganic compounds from a mixture.Is it possible to distinguish two spots that have the same Rf value?
5- Yes, it is possible to distinguish two spots that have the same Rf value but represent different compounds.What does a high RF value mean?
Definition. Rf = distance traveled by substance/distance traveled by solvent front. A high Rf (Ie 0.92) would refer to a substance that is very non-polar. Ie that substance moved a 92% of the entire distance the solvent traveled. A low Rf value (0.10) would refer to a substance that is very polar.What is the mobile and stationary phase in chromatography?
Chromatography is used to separate mixtures of substances into their components. They all have a stationary phase (a solid, or a liquid supported on a solid) and a mobile phase (a liquid or a gas). The mobile phase flows through the stationary phase and carries the components of the mixture with it.How is RF value calculated?
With your ruler, measure the distance the solvent traveled, which is Df, and measure the distance the test solution traveled, which is Ds. Calculate the retention factor using this equation: RF = Ds/Df. Simply divide the distance the solution traveled by the distance the solvent traveled.What does the RF value mean in TLC?
The retention factor, or Rf, is defined as the distance traveled by the compound divided by the distance traveled by the solvent. The larger an Rf of a compound, the larger the distance it travels on the TLC plate.On what factors Rf value depends?
factors which affect Rf value are:-• The solvent system and its composition. Temperature. The quality of the paper. Distance through which the solvent runs.