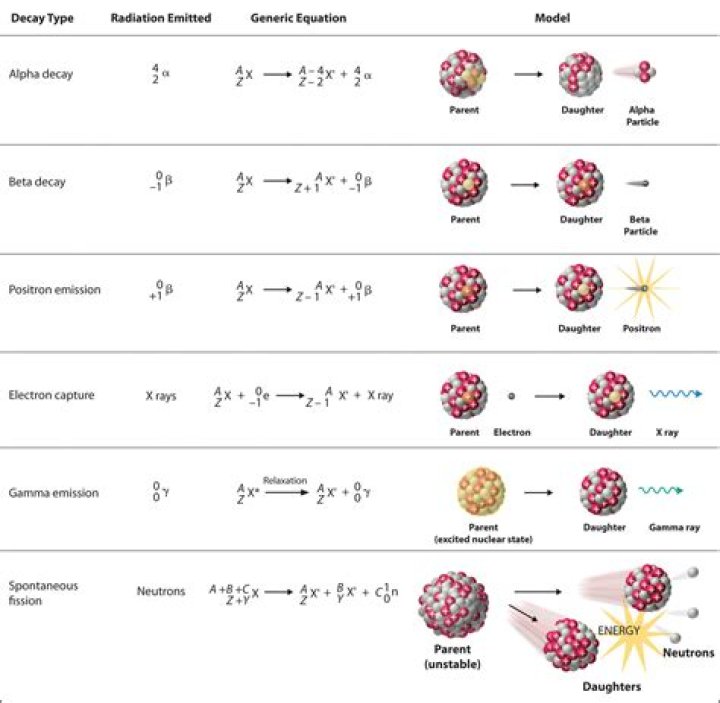
Technically speaking, alpha and beta decay are both types of nuclear fission. Fission is the breaking down of an atom's nucleus into smaller parts. This produces an element that is two protons smaller than the parent atom. Beta decay is the breaking down of a nucleus to produce a beta particle (high energy electron)..
Keeping this in view, is nuclear fission beta decay?
Fission products have, on average, about the same ratio of neutrons and protons as their parent nucleus, and are therefore usually unstable to beta decay (which changes neutrons to protons) because they have proportionally too many neutrons compared to stable isotopes of similar mass.
One may also ask, how do you do alpha and beta decay? Writing Alpha and Beta Decay Equations
- The nucleus of an atom splits into two parts.
- One of these parts (the alpha particle) goes zooming off into space.
- The nucleus left behind has its atomic number reduced by 2 and its mass number reduced by 4 (that is, by 2 protons and 2 neutrons).
Correspondingly, how are fission fusion and radioactive decay similar and different?
Nuclear fusion - two or more small nuclei combine at high speeds to form a single, larger nucleus. Two hydrogen nuclei fuse to form a helium nucleus. Fusion releases a TON of energy--more than fission and radioactive decay. Examples: The sun's energy comes from hydrogen atoms combining to make helium.
What characteristic of fission makes a chain reaction?
A fission chain reaction occurs when the fission of one atom produces enough neutrons to produce one or more other fission reaction(s) in the fissionable material, and that fission reaction does the same, etc.
Related Question Answers
What does U 235 decay into?
Uranium-235 Decay Chain The decay chain of this radioactive metal is known as the Actinium Series withThorium-231 being the next isotope in this decay process. It makes Thorium-231 the daughter nuclide of this isotope. Uranium-235 is also known as Actinouranium as it is the parent isotope of the Actinium Series.Who discovered nucleus?
Ernest Rutherford
What does radioactive decay produce?
Radioactive decay is the spontaneous breakdown of an atomic nucleus resulting in the release of energy and matter from the nucleus. Remember that a radioisotope has unstable nuclei that does not have enough binding energy to hold the nucleus together.What is fission used for?
Nuclear fission is a process whereby energy is released by the splitting of uranium atoms. Fission releases heat energy that can generate steam, which is used to spin a turbine to produce electricity. Nuclear Energy.Why is uranium used in fission?
The isotope U-235 is important because under certain conditions it can readily be split, yielding a lot of energy. It is therefore said to be 'fissile' and we use the expression 'nuclear fission'. Meanwhile, like all radioactive isotopes, they decay.Where is uranium found?
Uranium ore deposits are economically recoverable concentrations of uranium within the Earth's crust. Uranium is one of the more common elements in the Earth's crust, being 40 times more common than silver and 500 times more common than gold. It can be found almost everywhere in rock, soil, rivers, and oceans.Is natural uranium dangerous?
Because uranium is a radioactive substance health effects have been researched. Scientists have detected no harmful radiation effects of natural levels of uranium. However, chemical effects may occur after the uptake of large amounts of uranium and these can cause health effects such as kidney disease.How is fission started?
In order to initiate most fission reactions, an atom is bombarded by a neutron to produce an unstable isotope, which undergoes fission. When neutrons are released during the fission process, they can initiate a chain reaction of continuous fission which sustains itself.What is radioactive decay equation?
The radioactive decay law states that the probability per unit time that a nucleus will decay is a constant, independent of time. The radioactive decay of certain number of atoms (mass) is exponential in time. Radioactive decay law: N = N.e-λt. The rate of nuclear decay is also measured in terms of half-lives.Why is radioactive decay the opposite of nuclear fusion?
1 Answer. nuclear fusion is a nuclear reaction in which two or more atomic nuclei come very close and then collide at a very high speed and join to form a new nucleus. Radioactive decay is the breakdown of an atomic nucleus resulting in the release of energy and matter from the nucleus.What type of decay is nuclear fission?
Fission is a radioactive decay mode that is only common in quite heavy nuclei. Especially if it happens spontaneously, fission is really just a special mode of radioactive decay, in which a large nucleus splits into one or more fragments.Is the sun a nuclear reactor?
The sun is the best nuclear fusion reactor around. NASA Goddard Spaceflight Center Quite simply: Gravity. Fusion in the sun's center works by pressing four hydrogen atoms together to form one helium atom and energy. The extreme weight of the sun provides the pressure needed.What is the equation for nuclear fusion?
The binding energy B is the energy associated with the mass difference between the Z protons and N neutrons considered separately and the nucleons bound together (Z + N) in a nucleus of mass M. The formula is B = (Zmp + Nmn − M)c2, where mp and mn are the proton and neutron masses and c is the speed of light.How do you know if its fission or fusion?
Both fission and fusion are nuclear reactions that produce energy, but the applications are not the same. Fission is the splitting of a heavy, unstable nucleus into two lighter nuclei, and fusion is the process where two light nuclei combine together releasing vast amounts of energy.Why is nuclear fusion good?
The following advantages make fusion worth pursuing. Abundant energy: Fusing atoms together in a controlled way releases nearly four million times more energy than a chemical reaction such as the burning of coal, oil or gas and four times as much as nuclear fission reactions (at equal mass).Is gamma decay fission or fusion?
Gamma-rays are produced primarily by four different nuclear reactions: fusion, fission, alpha decay and gamma decay. Nuclear fusion is the reaction that powers the sun and stars.What is alpha decay used for?
Alpha decay can provide a safe power source for radioisotope thermoelectric generators used for space probes and were used for artificial heart pacemakers. Alpha decay is much more easily shielded against than other forms of radioactive decay.Is alpha decay harmful to humans?
Biological effects Due to the short range of absorption and inability to penetrate the outer layers of skin, alpha particles are not, in general, dangerous to life unless the source is ingested or inhaled.What is a beta particle made of?
Alpha radiation consists of helium nuclei and is readily stopped by a sheet of paper. Beta radiation, consisting of electrons or positrons, is stopped by thin aluminum plate, but gamma radiation requires shielding by dense material such as lead, or concrete.