How is cyclohexene prepared from cyclohexanol?
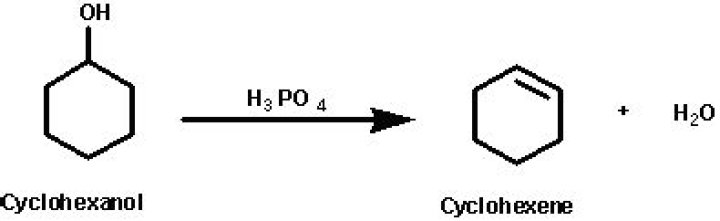
.
People also ask, what type of reaction is the conversion of cyclohexanol to cyclohexene?
E2 stands for bimolecular elimination, a one-step mechanism that breaks carbon hydrogen and carbon halogen bonds to form a double bond. The E2 mechanism is very similar to the SN2 reaction mechanism. Protonation of cyclohexanol produces an oxonium ion, which then reacts with water to form cyclohexene.
Beside above, how do you purify cyclohexene? Add a few boiling stones to the flask, and purify the product by simple distillation. Use a tared vial as a receiver, and cool the receiver in an ice bath. Collect distillate with a head temperature between 80-85 °C. Obtain a weight of your purified product.
Subsequently, question is, how do you find the cyclohexanol theoretical yield of cyclohexene?
Convert this number of moles of cyclohexene to grams of cyclohexene by multiplying by the MW of cyclohexene (82.1 g/mol). In other words, 2.05 g of cyclohexanol should produce 1.68 g of cyclohexene. This is the best-case yield also known as the theoretical yield.
What is cyclohexene used for?
Cyclohexene is a clear, colorless liquid with a sweet odor. It is used in oil extraction, to make other chemicals, and as a catalyst solvent. It is also found as a component of motor vehicle exhaust.
Related Question AnswersHow can you distinguish between cyclohexene and hexene?
1. Cyclohexane and cyclohexene both are cyclic compounds. Cyclohexane is an alkane meaning it only contains a single bond while cyclohexene contains a double bond. Both have different IR absorption bands.Why cyclohexene has no isomers?
No. Cyclohexene is too small of a ring to exhibit the trans isomer. Trying to force it to be trans would place a massive amount of ring strain on the molecule. This is true for all rings with an alkene up to cyclooctene.Why is cyclohexene insoluble in water?
All the bonds are C-H with the exception of one C=C double bond. The polarity of these bonds is very weak. So the molecule is largely non polar. Thus a polar substance like water will not be miscible with a non polar substance like cyclohexene as it is very difficult to form intermolecular bonds between them.What Colour is cyclohexene?
Cyclohexene| Names | |
|---|---|
| Chemical formula | C6H10 |
| Molar mass | 82.143 g/mol |
| Appearance | colorless liquid |
| Odor | sweet |
Is cyclohexene a alkene?
Description: Cyclohexene is a colorless liquid (cyclic alkene) with a sweetish odor.Is cyclohexene flammable?
Cycloolefin. Cyclohexene is a cyclic olefin, and is a colorless, flammable liquid with a special pungent odor at room temperature.What is the difference between cyclohexene and benzene?
is that benzene is (organic compound) an aromatic hydrocarbon of formula c6h6 whose structure consists of a ring of alternate single and double bonds while cyclohexene is (organic compound) a cyclic hydrocarbon containing six carbon atoms and one double bond; any of its derivatives.What type of alcohol is cyclohexanol?
Cyclohexanol is an alcohol that consists of cyclohexane bearing a single hydroxy substituent. The parent of the class of cyclohexanols. It has a role as a solvent. It is a secondary alcohol and a member of cyclohexanols.What is the role of cyclohexanol in this reaction?
What is the role of cyclohexanol in this reaction? It gets reduced and it acts as the reducing agent. It gets reduced and it acts as the oxidizing agent. It gets oxidized and it acts as the reducing agent.What kind of hydrocarbon is cyclohexene?
In the reactions we will perform in this experiment, hexane will be used to represent the saturated hydrocarbons, cyclohexene will be used as an unsaturated hydrocarbon, and toluene, the aromatic hydrocarbon.What is the most commonly used dehydrating agent?
Chemicals commonly used as dehydrating agents include concentrated phosphoric acid, concentrated sulfuric acid, hot ceramic and hot aluminum oxide. A dehydration reaction may also be known as a condensation reaction, but more properly, a dehydration reaction is a specific type of condensation reaction.What is the purpose of dehydration of cyclohexanol?
The dehydration of cyclohexanol to give cyclohexene Cyclohexanol is heated with concentrated phosphoric(V) acid and the liquid cyclohexene distils off and can be collected and purified. Phosphoric(V) acid tends to be used in place of sulphuric acid because it is safer and produces a less messy reaction.Why cyclohexane and cyclohexene is insoluble in water?
Cyclohexene is a non polar molecule and water is polar molecule as it has sufficient electronegativity difference between the constituent atoms. So cyclohexene won't be aoluble in water. Instead it will be soluble in non polar solvents like benzene.Why is the boiling point of cyclohexanol higher than cyclohexene?
Cyclohexanol molecules have a strong attraction to each other due to the hydrogen bonding between alcohol groups in these molecules. Boiling point of a compound is directly related to intermolecular force strength which means cyclohexanol is going to have a higher boiling point than cyclohexene.Why is Methylcyclohexene a major product?
Dehydration of an alcohol removes the OH and the H on the β-carbon. Dehydration of an alcohol gives the more stable alkene (more highly substituted) as the major product. The major product is 1-methylcyclohexene and methylenecyclohexane is the minor product.How do you turn cyclohexene into cyclohexene?
Commercially, cyclohexene is made from hydrogenation of benzene. If you want a lab prep from cyclohexane, the first step would be reaction with chlorine or bromine to give C6H11X (X = halogen). Then elimination with a bulky base like potassium tert-butoxide gives cyclohexene.What is the theoretical yield of cyclohexene?
Convert this number of moles of cyclohexene to grams of cyclohexene by multiplying by the MW of cyclohexene (82.1 g/mol). In other words, 2.05 g of cyclohexanol should produce 1.68 g of cyclohexene. This is the best-case yield also known as the theoretical yield.How do I calculate theoretical yield?
How to Calculate Theoretical Yields- Determine the number of moles of each reactant.
- Multiply the molecular weight by the number of moles in the equation.
- Calculate the theoretical mole yield by using the chemical equation.
- Multiply the number of moles of the product by the molecular weight of the product to determine the theoretical yield.



