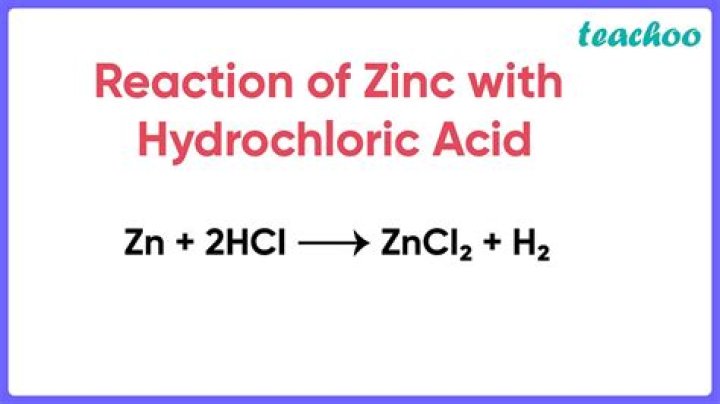
The metal zinc readily reacts with hydrochloric acid to produce hydrogen gas (H2) and zinc chloride (ZnCl2). The zinc reaction produces the heat and hence has the negative enthalpy..
In this manner, what happens when zinc reacts with hydrochloric acid?
It is a single replacement reaction where zinc metal displaces the hydrogen to form hydrogen gas and zinc chloride, a salt. Zinc reacts quickly with the acid to form bubbles of hydrogen. Explanation: the metal zinc reacts with hydrochloric acid it produce hydrogen gas and zinc chloride.
Beside above, what happens when dilute HCl is added to zinc pieces? Zinc reacts with dilute hydrochloric acid to form zinc chloride and hydrogen gas. Hydrogen gas will be seen as bubbles around the zinc granules. zinc chloride is formed and hydrogen gas is released.
Subsequently, one may also ask, does zinc dissolve in hydrochloric acid?
Yes, zinc (Zn) dissolves in hydrochloric acid (HCl). Zinc is more reactive than hydrogen, as the reactivity series says. Therefore, zinc can displace hydrogen from the HCl and form its soluble chloride, that is, zinc chloride (ZnCl2).
Does Zinc react with NaOH?
Zinc is a transition metal which shows moderate reactivity. Zinc reacts with NaOH to form sodium zincate with the evolution of hydrogen gas.
Related Question Answers
What type of reaction is zinc oxide and hydrochloric acid?
Zinc oxide reacts with hydrochloric acid to form zinc chloride and water. Zinc oxide is insoluble in water.Why does Zinc react slowly with hydrochloric acid?
Explain why zinc reacts more slowly with dilute hydrochloric acid than with concentrated hydrochloric acid? This allows more zinc atoms per second to be ionized (reducing the hydrogen in the process) and dissolve into the solution.What happens when magnesium reacts with hydrochloric acid?
When magnesium reacts with hydrochloric acid, a gas is released. When magnesium reacts with hydrochloric acid, hydrogen gas is released.Why does zinc turn black in hydrochloric acid?
Extremely finely ground metals appear black. The formation of hydrogen gas may form little pockets in the zinc as it is dissolving.What salt is formed when zinc reacts with hydrochloric acid?
zinc chloride
How do you make hydrogen with zinc and hydrochloric acid?
Zinc also reacts with hydrochloric acid to produce zinc chloride and hydrogen. Sulfuric acid or hydrochloric acid can be mixed with water to any levil of dilution. Pour the acid slowly into the water. Do not pour the water into the acid.What happens when zinc reacts with nitric acid?
The products formed when zinc reacts with dilute nitric acid are zinc nitrate and hydrogen gas; i.e. 5Zn + 12HNO3 ----> 5Zn(NO3)2 + N2 + 6H2O. These products are formed due to the reduction of HNO3 by nascent hydrogen, which is produced in increasing amounts as the concentration of nitric acid is lowered; e.g.What is the Colour of zinc granules?
WHAT IS THE COLOUR AND SHAPE OF ZINC GRANULES ? they are grey in colour and does not have any regular shape. They will not be formed on the zinc granules. They'll come out as deposits.What happens when iron reacts with hydrochloric acid?
When dilute hydrochloric acid is added to iron hydrogen gas and iron(II)chloride are produced. Iron is said to displace hydrogen ions in solution producing fizzing colourless odourless hydrogen gas which pops near flame and iron(II)chloride are produced which is pale green is colour.How do you dissolve zinc?
Zinc is insoluble in water but reacts readily with non-oxidising acids, forming zinc (II) and releasing hydrogen. It also dissolves in strong bases. It reacts readily when heated with oxygen to give zinc oxide. Direct reactions with halogens will also occur.Does nitric acid dissolve zinc?
Aluminum and zinc do not react with concentrated nitric acid since a thick, hard to dissolve oxidation layer builds (passive oxidation) that protects the metal against further assaults. Zinc, however, reacts with diluted nitric acid (for an analog, see Experiment 4.4. 24.1).What does sodium hydroxide and hydrochloric acid make?
Hydrochloric acid reacts with sodium hydroxide to form sodium chloride (the salt) and water.Why does Zinc react with hydrochloric acid and not copper?
1 Answer. Zinc reacts with hydrochloric acid to produce hydrogen gas because it's more reactive than hydrogen, and thus displaces the latter from an acid. By comparison, copper cannot displace hydrogen from HCl because it's less reactive than hydrogen. Zinc will reduce the hydrogen and get oxidized in the process.Does magnesium react with hydrochloric acid?
Magnesium reacts with hydrochloric acid according to the equation: Mg(s) + 2 HCl(aq) --> MgCl 2(aq) + H 2(g) This demonstration can be used to illustrate the characteristic reaction of metals with acid, a single replacement reaction, or to demonstrate the generation of hydrogen gas.What happens when copper reacts with hydrochloric acid?
There will be no reaction. Copper is a very unreactive metal, and it does not react with hydrochloric acid. It is above copper in a metal reactivity series, so copper cannot replace the hydrogen in HCl to form CuCl2 .What is the observation when a small piece of zinc is added to dilute hydrochloric acid?
Zinc reacts quickly with the acid to form bubbles of hydrogen. ♠When zinc granules are added to dilute hydrochloric acid, then zinc chloride is formed along the evolution of hydrogen gas .What is observed when dilute hydrochloric acid is added to zinc metal in a test tube?
Zinc chloride and hydrogen gas is form which is highly unstable so by hydronium process it combined by water . The Brainliest Answer! If we adds hcl to zinc metal the zinc displaces the hydrogen and form Zncl2 and h2 gas is evolve.What would you observe on adding dilute HCl to zinc metal in a test tube?
When dilute hydrochloric acid is added to granulated zinc placed in a test tube, a colourless and odourless gas is evolved with bubbles.What happens when dilute HCl is added to baking soda?
What happen when dilute hydrochloric acid is added to baking soda? baking soda will reacts with HCl to form sodium chloride, water and carbon dioxide. Explanation: (ii) Baking soda reacts with HCl to form sodium chloride, water and carbon dioxide.