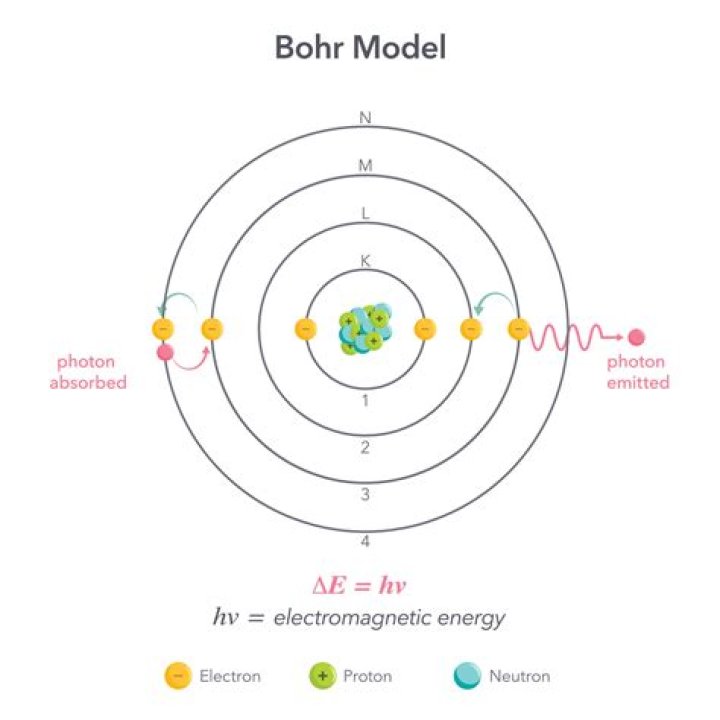
Niels Bohr explained the line spectrum of the hydrogen atom by assuming that the electron moved in circular orbits and that orbits with only certain radii were allowed. This produces an absorption spectrum, which has dark lines in the same position as the bright lines in the emission spectrum of an element..
Keeping this in view, how is hydrogen's emission spectrum produced according to Bohr?
An excited state is a higher energy level. According to Bohr's model of the hydrogen atom, how is hydrogen's emission spectrum produced? When an electron falls from a higher to a lower orbit (energy level) energy is given off (orbit). The different orbits produce fixed lines in the emission spectrum of hydrogen.
Subsequently, question is, what is the Bohr model of the atom and how does it explain both emission and absorption spectra? Absorption and emission According to Bohr's model, an electron would absorb energy in the form of photons to get excited to a higher energy level as long as the photon's energy was equal to the energy difference between the initial and final energy levels.
In this manner, how does the Bohr model explain emission spectra?
Bohr's atomic model hydrogen emission spectra. Bohr explained that electrons can be moved into different orbits with the addition of energy. When the energy is removed, the electrons return back to their ground state, emitting a corresponding amount of energy – a quantum of light, or photon.
What is the hydrogen emission spectrum?
The high voltage in a discharge tube provides that energy. Hydrogen molecules are first broken up into hydrogen atoms (hence the atomic hydrogen emission spectrum) and electrons are then promoted into higher energy levels. Suppose a particular electron was excited into the third energy level.
Related Question Answers
How can you use the emission spectrum of an element to identify it?
Each natural element has a characteristic light spectrum that helps identify it in samples of unknown substances. Spectroscopy is the practice of examining spectra and comparing them to those of known elements. Using spectroscopy methods, scientists can identify pure substances or compounds and the elements in them.Why are only 4 lines seen in the hydrogen emission spectrum?
Based on the wavelengths of the spectral lines, Bohr was able to calculate the energies that the hydrogen electron would have in each of its allowed energy levels. He found that the four visible spectral lines corresponded to transitions from higher energy levels down to the second energy level (n = 2).How do you read a Bohr model?
- Draw the nucleus.
- Write the number of neutrons and the number of protons in the nucleus.
- Draw the first energy level.
- Draw the electrons in the energy levels according to the rules below.
- Keep track of how many electrons are put in each level and the number of electrons left to use.
What are sublevels?
A sublevel is an energy level defined by quantum theory. In chemistry, sublevels refer to energies associated with electrons. In physics, sublevels may also refer to energies associated with the nucleus. Shell 4 can hold up to 32 electrons, Shell 5 can hold up to 50 electrons,How is an emission spectrum produced?
Thus, emission spectra are produced by thin gases in which the atoms do not experience many collisions (because of the low density). The emission lines correspond to photons of discrete energies that are emitted when excited atomic states in the gas make transitions back to lower-lying levels.Why is Bohr's model incorrect?
2 Answers. The main problem with Bohr's model is that it works very well for atoms with only one electron, like H or He+, but not at all for multi-electron atoms. Bohr's model allows classical behavior of an electron (orbiting the nucleus at discrete distances from the nucleus.What is Bohr equation?
Bohr equation. The Bohr equation, named after Danish physician Christian Bohr (1855–1911), describes the amount of physiological dead space in a person's lungs. This is given as a ratio of dead space to tidal volume. It differs from anatomical dead space as measured by Fowler's method as it includes alveolar dead spaceHow many energy levels are there in hydrogen?
The formula defining the energy levels of a Hydrogen atom are given by the equation: E = -E0/n2, where E0 = 13.6 eV (1 eV = 1.602×10-19 Joules) and n = 1,2,3… and so on. The energy is expressed as a negative number because it takes that much energy to unbind (ionize) the electron from the nucleus.Why is the Bohr model important?
The Bohr Model of the atom, introduced in 1913 by Niels Bohr, is extremely important. The Bohr Model explains to us that electrons or negative charges orbit around the atom's nucleus in energy levels. It also describes that electrons can change energy levels.What are the four principles of Bohr's model?
The Bohr model can be summarized by the following four principles: Electrons occupy only certain orbits around the nucleus. Those orbits are stable and are called "stationary" orbits. Each orbit has an energy associated with it.Why does Bohr model only work for hydrogen?
The Bohr model works only for hydrogen because it considers only the interactions between one electron and the nucleus. The Bohr model is based on the energy levels of one electron orbiting a nucleus at various energy levels. Any other electrons in the atom will repel the one electron and change its energy level.What happens to an electron when energy is added?
When an electron absorbs energy, it jumps to a higher orbital. An electron in an excited state can release energy and 'fall' to a lower state. When it does, the electron releases a photon of electromagnetic energy. The electron can absorb one quantum of energy and jump up to the excitation state.What is the texture of hydrogen?
Hydrogen is a colorless, odorless gas which exists, at standard temperature and pressure, as diatomic molecules, H2. The biliong point of hydrogen is -252,87 0 C.What was the major failure of the Bohr model of the atom?
The Bohr model failed because it failed to take into account one thing: synchrotron radiation. The Bohr model says that electrons whizz around the atomic nucleus in the same way that the earth goes around the sun - but rather than being bound by gravity, it is bound by electromagnetic forces.How did Bohr discover energy levels?
In 1913, Niels Bohr proposed a theory for the hydrogen atom based on quantum theory that energy is transferred only in certain well defined quantities. Electrons should move around the nucleus but only in prescribed orbits. When jumping from one orbit to another with lower energy, a light quantum is emitted.What do absorption lines tell us?
As photons fly through the outermost layers of the stellar atmosphere, however, they may be absorbed by atoms or ions in those outer layers. The absorption lines produced by these outermost layers of the star tell us a lot about the chemical compositition, temperature, and other features of the star.How do you tell if a photon is emitted or absorbed?
The atom absorbs or emits light in discrete packets called photons, and each photon has a definite energy. Only a photon with an energy of exactly 10.2 eV can be absorbed or emitted when the electron jumps between the n = 1 and n = 2 energy levels. l = hc/E.What is the difference between emission and absorption spectra?
The difference between absorption and emission spectra are that absorption lines are where light has been absorbed by the atom thus you see a dip in the spectrum whereas emission spectra have spikes in the spectra due to atoms releasing photons at those wavelengths.What are two limitations of the Bohr model for the atom?
The Bohr Model is very limited in terms of size. Poor spectral predictions are obtained when larger atoms are in question. It cannot predict the relative intensities of spectral lines. It does not explain the Zeeman Effect, when the spectral line is split into several components in the presence of a magnetic field.