How does steric hindrance effect acidity?
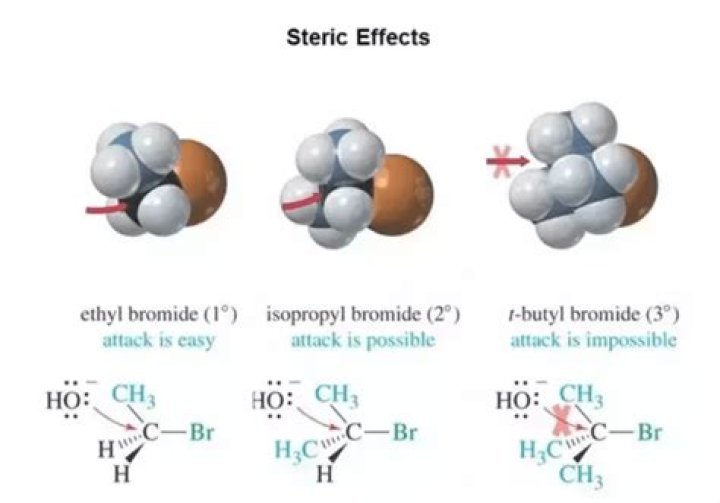
.
Also asked, what causes steric hindrance?
Steric Hindrance. Steric hindrance at a given atom in a molecule is the congestion caused by the physical presence of the surrounding ligands, which may slow down or prevent reactions at the atom.
Secondly, what is meant by steric effect? Steric effects are the effects seen in molecules that come from the fact that atoms occupy space. When atoms are put close to each other, this costs energy. The electrons near the atoms want to stay away from each other. This can change the way molecules want to react. An example of steric effects is steric hindrance.
Regarding this, what is meant by steric hindrance in chemistry?
Steric hindrance is the stopping of a chemical reaction which might be caused by a molecule's structure. An example of steric hindrance is how rotaxanes are created. YourDictionary definition and usage example. Copyright © 2018 by LoveToKnow Corp. MLA Style.
Does steric hindrance increase stability?
Increased steric hindrance around phosphorous atoms in phosphites increases their hydrolytic stability. and therefore cannot attack polymer chains. Steric hindrance of ortho-position to the hydroxyl group affects the radical reaction rate.
Related Question AnswersWhat is resonance effect?
Resonance effect is the polarity produced in a molecule due to interaction between a lone pair of electron and a pi bond or it is produced due to interaction of two pi bonds between two adjacent atoms.How does steric hindrance effect sn2?
How does steric hindrance affect the rate at which an SN 2 reaction will occur? As each hydrogen is replaced by an R group, the rate of reaction is significantly diminished. This is because the addition of one or two R groups shields the backside of the electrophilic carbon, impeding nucleophilic attack.Why does steric hindrance occur?
Steric hindrance or steric resistance occurs when the size of groups within a molecule prevents chemical reactions that are observed in related smaller molecules. Steric hindrance between adjacent groups can also restrict torsional bond angles.How do you find a steric number?
The steric number is calculated using the following formula: Steric Number = (number of lone electron pairs on the central atom) + (number of atoms bonded to the central atom)What is steric acceleration?
The steric effect in a reaction is ascribed to the difference in steric energy between, on the one hand, reactants and, on the other hand, a. transition state. (or products). A steric effect on a rate process may result in a rate increase ('steric. acceleration.What is torsional strain?
Torsional strain or eclipsing strain is the increase in potential energy of a molecule due to repulsion between electrons in bonds that do not share an atom. eg: Consider two conformations of ethane: The smallest dihedral angle is 60º in 1; it is 0º in 2. Consequently, torsional strain is greater in 2 than in 1.What is Saytzeff rule in organic chemistry?
Zaitsev's rule (or Saytzeff's rule, Saytzev's rule) is an empirical rule for predicting the favored alkene product(s) in elimination reactions. More generally, Zaitsev's rule predicts that in an elimination reaction, the most substituted product will be the most stable, and therefore the most favored.What is steric effect with example?
Steric effects are the effects seen in molecules that come from the fact that atoms occupy space. An example of steric effects is steric hindrance. This is when a large group in a molecule makes reactions not work. For example, an SN2 reaction does not happen on carbon atoms that have three substituents.What makes a strong Nucleophile?
Charge. “The conjugate base is always a better nucleophile”. HO- is a better nucleophile than H2O. The greater the negative charge, the more likely an atom will give up its pair of electrons to form a bond.What is Electrophile and Nucleophile?
Electrophile and nucleophile are the chemical species that donate or accept electrons to form a new chemical bond. Any molecule, ion or atom that is in some manner deficient in electron can act as an electrophile. A nucleophile is usually charged negatively or neutral with a lone couple of donable electrons.What is inductive effect in chemistry?
In chemistry, the inductive effect is an effect regarding the transmission of unequal sharing of the bonding electron through a chain of atoms in a molecule, leading to a permanent dipole in a bond.How do you find the inductive effect?
Basically the inductive effect is just the slight push or pull of electron density arising due to electronegativity difference. So next time if you want to check I effect just find out whether there exist electronegativity difference or not.What is positive and negative inductive effect?
-I effect:— Electron withdrawing substituents tend to attract electron pair,due to which carbon to which substituent is attached,carries positive charge. This effect is called as negative inductive effect and the groups which cause this effect are called as -I groups.What is inductive effect explain with example?
An inductive effect is an electronic effect due to the polarisation of σ bonds within a molecule or ion. Positive inductive effect refers to electron releasing tendency of functional groups. For example, alkyl, aryl, metals, etc. Negative inductive effect refers to electron accepting tendency of functional groups.What is +i and effect in chemistry?
+I Effect (Positive Inductive Effect) When a chemical species with the tendency to release or donate electrons, such as an alkyl group, is introduced to a carbon chain, the charge is relayed through the chain and this effect is called the Positive Inductive Effect or the +I Effect.Is E or Z more stable?
E/Z notationEdit Z (from the German zusammen) means together and usually corresponds to the term cis; E (from the German entgegen) means opposite and usually corresponds to the term trans. Usually, E isomers are more stable than Z isomers because of steric effects.Which alkene is most stable?
Since, the most alkyl groups are attached in 3-methylpent-2-ene among all the given alkenes, the most stable alkene among the given alkenes is 3-methylpent-2-ene.Which alkene is more reactive?
Alkenes are relatively stable compounds, but are more reactive than alkanes because of the reactivity of the carbon–carbon π-bond. Most reactions of alkenes involve additions to this π bond, forming new single bonds. The carbon-carbon double bond in alkenes such as ethene react with concentrated sulfuric acid.How do you determine if a molecule is stable?
These rules are in approximate order of importance.- Octet rule. Having 8 valence electrons around an atom (2 in the case of H) is a particularly stable arrangement.
- Minimize formal charge.
- Negative on Electronegative.
- Resonance stabilizes.
- Inductive effects.
- 3-dimensional arrangement of atoms influences stability.



