How does AAS relate to absorbance spectra?
.
Also question is, how does the AAS work?
Atomic absorption spectrometers use the absorption of light to measure the concentration of gas phase atoms. A high voltage is passed between the cathode and anode and the metal atoms are excited into producing light with a certain emission spectrum.
Subsequently, question is, what does AAS stand for in chemistry? Atomic absorption spectroscopy
Similarly, it is asked, does AAS follow Beer's law?
Atoms of an element emit a characteristic spectral line. However, applying the Beer-Lambert law directly in AAS is difficult due to: variations in atomization efficiency from the sample matrix, non-uniformity of concentration and path length of analyte atoms (in graphite furnace AA).
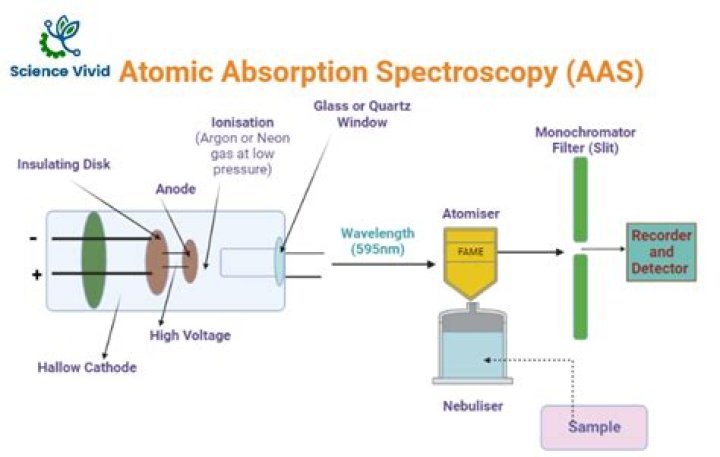
What is the working principle of AAS?
The technique uses basically the principle that free atoms (gas) generated in an atomizer can absorb radiation at specific frequency.? Atomic-absorption spectroscopy quantifies the absorption of ground state atoms in the gaseous state .? The atoms absorb ultraviolet or visible light and make transitions to
Related Question AnswersWhy are hollow cathode lamps used in AAS?
Typical Hollow Cathode Lamp Setup Due to creation of plasma from the inert gas, sputtering of atoms from the cathode takes place as inert gas ions get bombarded on the cathode. The excited atoms of the inert gas and cathode are used for detection and to measure the spectrum while testing.Is AAS qualitative or quantitative?
Atomic absorption spectroscopy (AAS) is a quantitative spectro-analytical procedure for the determination of chemical elements using the absorption of optical radiation by free atoms in the gaseous state (Welz and Sperling, 2008).What is the purpose of flame in AAS?
Flame Atomic Absorption Spectrometry Flame Atomic Absorption is a very common technique for detecting metals present in samples. The technique is based on the principle that ground state metals absorb light at a specific wavelength. Metal ions in a solution are converted to atomic state by means of a flame.Why Acetylene is used in AAS?
In FAAS, the most common AA technique, Acetylene is commonly used as a fuel gas and requires low levels of Phosphine and Hydrogen Sulphide to give a cleaner flame and less interference to ensure optimum analytical accuracy.What is the purpose of Atomic Absorption Spectroscopy?
Atomic Absorption Spectroscopy (AAS) Atomic-absorption (AA) spectroscopy uses the absorption of light to measure the concentration of gas-phase atoms. Since samples are usually liquids or solids, the analyte atoms or ions must be vaporized in a flame or graphite furnace.How do you find a sample concentration in AAS?
Using dilution equation (formula):- c1V1 = c2V2 c1 = lead concentration in undiluted sample = ? ppm. V1 = volume of undiluted sample in litres = 10/1000 = 0.010 L.
- Substitute the values into the equation and solve: c1 x 0.010 = 1.60 x 0.100. c1 x 0.010 = 0.16. c1 = 0.16 ÷ 0.010.
What is interference in AAS?
An interference is a phenomenon that affects the measurement or the population of ground state atoms of an analyte element. Related links: Flame Atomic Absorption. Cold vapor Hg/As. Interferences in AA analysis.What is the principle of AAS?
Principle of AAS AAS quantitatively measures the concentrations of elements present in a liquid sample. It utilises the principle that elements in the gas phase absorb light at very specific wavelengths which gives the technique excellent specificity and detection limits.What is Beer's law in chemistry?
Beer's Law is an equation that relates the attenuation of light to properties of a material. The law states that the concentration of a chemical is directly proportional to the absorbance of a solution.What is characteristic concentration in AAS?
Characteristic Concentration The ''characteristic concentration'' ( sometimes called ''sensitivity'') is a convention for defining the magnitude of the absorbance signal which will be produced by a given concentration of analyte.What is background absorption?
When the magnetic field is off, both the atomic and the background absorption occurs. When the magnetic field is on, only background absorption occurs. Again, the background signal is subtracted from the combined atomic and background signal to obtain a net atomic absorption signal.What kind of information do you get from atomic absorption spectroscopy?
Atomic absorption spectrometry (AAS) detects elements in either liquid or solid samples through the application of characteristic wavelengths of electromagnetic radiation from a light source. Individual elements will absorb wavelengths differently, and these absorbances are measured against standards.What is the chemical name of AS?
The elements of the periodic table sorted by name in an alphabetical list.| Name chemical element | Symbol | Atomic number |
|---|---|---|
| Argon | Ar | 18 |
| Arsenic | As | 33 |
| Astatine | At | 85 |
| Barium | Ba | 56 |



