How do you remove salt from sea sand?
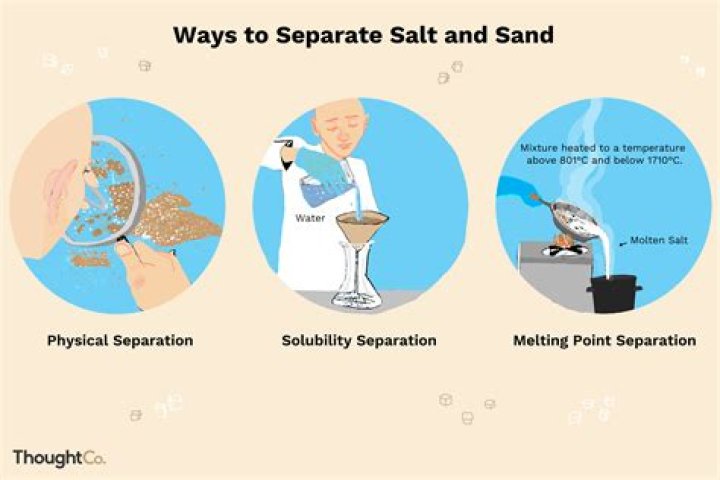
- Pour the salt and sand mixture into a pan.
- Add water.
- Heat the water until the salt dissolves.
- Remove the pan from heat and allow it to cool until it'ssafe to handle.
- Pour the salt water into a separate container.
- Now collect the sand.
- Pour the salt water back into the empty pan.
.
Beside this, is there salt in sand?
Beaches contain salt water and sand,therefore, naturally, the salt contains salt aswell.
Similarly, how do you clean beach sand? Steps
- Gather twice as much sand as you'll need. You'll likely losesome sand during the cleaning process.
- Sift sand to remove unwanted rocks and debris.
- Rinse out organic matter and other unwanted particles.
- Sterilize beach sand by baking it.
- Remove salt from beach sand by simmering it with water.
In respect to this, how would you separate iron sand and salt?
Water Soluble Iron is magnetic and the other two not, whichmeans a magnet could be used to attract the iron filings outof the mixture, leaving the salt and sand.Salt is water soluble, while sand is not. This meansthe two can be mixed in water and stirred. The salt willdissolve and the sand will not.
What method is used to separate sand and water?
When sand is added to water it eitherhangs in the water or forms a layer at the bottom of thecontainer. Sand therefore does not dissolve in waterand is insoluble. It is easy to separate sand and water byfiltering the mixture. Salt can be separated from a solutionthrough evaporation.
Related Question AnswersHow do you separate sodium chloride and sand?
A mixture of sodium chloride and sand inwater can be separated by the process of filtration and then by theprocess of distillation.How do you dissolve sand?
Gently shake the container to dissolve thepowder. Open the container and pour the sand into the acidmixture. Observe the sand for 24 hours. The sand willdissolve, adding the composite minerals to the water andneutralizing the acid.Does beach sand contain salt?
Sea sand does not have high compressive strength,high tensile strength etc. so it cannot be used in constructionactivities. In addition to this, the salt in sea sandtends to absorb moisture from atmosphere, bringingdampness.Can a mixture of salt and sugar be separated by filtration?
Eg: Water is a pure substance made up of only watermolecules. 4] Can a mixture of salt and sugar be separated byfiltration? Ans: Filtration is a method that is used toseparate insoluble solids from a liquid. Since salt andsugar both dissolve in water, filtration cannot be usedto separate the two.How would you separate a mixture of salt and sugar?
Sugar can dissolve in alcohol but saltcan't dissolve in alcohol so we can add the mixture inalcohol and filter it and get sugar alcohol solution asseparate and salt is left over at theend.What happens when salt is dissolved in water?
Salt (sodium chloride) is made from positivesodium ions bonded to negative chloride ions. Water candissolve salt because the positive part of watermolecules attracts the negative chloride ions and the negative partof water molecules attracts the positive sodiumions.Does Himalayan salt have calcium?
Himalayan salt often contains trace amounts ofiron oxide (rust), which gives it a pink color. It alsohas small amounts of calcium, iron, potassium andmagnesium, making it slightly lower in sodium than regular tablesalt. Many people prefer the flavor of Himalayan saltover other types.How do you separate sugar and sand?
Sand does not dissolve in water but sugaris soluble in water. So to separate mixture of sand andsugar we dissolve it in water. The sugar dissolves inwater leaving behind sand. form of solution.How do you separate charcoal and sand?
Take the sand and charcoal mixture anddunk it into a container of water. The charcoal will floatout of the mixture and can be skimmed off the top and dried. Filterthe sand/water mixture to separate the sandand dry that too. You should be left with the 3 separatedsubstances of salt, sand and charcoal..How do you separate sawdust and sand?
Most likely any sand you have will be denser thanwater, so it will sink, and the sawdust is going to be lessdense than water, so it should float. mix it with a large amount ofwater, then scrape the sawdust off the top.- How do you separate sawdust and salt?
- Which method will we use to separate sawdust, sand, andsalt?
How do you separate common salt?
Separate Salt and Water UsingDistillation You can boil or evaporate the water and the saltwill be left behind as a solid. If you want to collect the water,you can use distillation. This works because salt has a muchhigher boiling point than water.How would you separate sand and gravel?
How to Separate Sand & Gravel- Lay a piece of plastic or tarp on the ground.
- Find a heavy-duty wire mesh screen.
- Place the screen over several buckets or a makeshift frame tokeep the screen off the ground.
- Pour the gravel and sand mixture on top of the screen.
- Remove the larger pieces of gravel and place them where youwant them.



