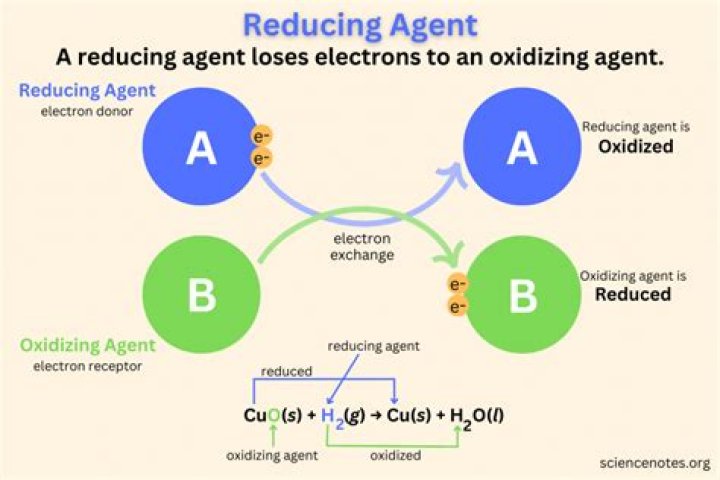
Reducing agents can be ranked by increasing strength by ranking their reduction potentials. The reducing agent is stronger when it has a more negative reduction potential and weaker when it has a more positive reduction potential..
In respect to this, how do you identify oxidizing and reducing agents?
A reducing agent is a substance that causes another substance to reduce. So to identify an oxidizing agent, simply look at the oxidation number of an atom before and after the reaction. If the oxidation number is greater in the product, then it lost electrons and the substance was oxidized.
Secondly, which elements are good reducing agents? Good reducing agents include the active metals, such as sodium, magnesium, aluminum, and zinc, which have relatively small ionization energies and low electro-negativities. Metal hydrides, such as NaH, CaH2, and LiAlH4, which formally contain the H- ion, are also good reducing agents.
In respect to this, how do you find the weakest reducing agent?
Explanation: Here's a typical table of standard reduction potentials. The species at the top left have the greatest "potential" to be reduced, so they are the strongest oxidizing agents. The strongest oxidizing agent in the list is F2 , followed by H2O2 , and so on down to the weakest oxidizing agent, Li+ .
Is HCl a strong reducing agent?
HCl (Cl^-) can be oxidized to chlorine gas (Deacon reaction) using a stronger oxidizing agent than HCl (TiO2). HCl (H^+) can be reduced to hydrogen gas via a weaker oxidizing agent than HCl (stronger reducing agent) such as Zn. There is no ultimate oxidizing agent or reducing agent.
Related Question Answers
What is a good oxidizing agent?
This means that they easily attract electrons. As you can see in the periodic table of elements, the halogens that are good oxidizing agents are fluorine, chlorine, bromine and iodine, with fluorine being the strongest oxidizing agent among the four, followed by chlorine, bromine and iodine.What is a reducing agent example?
A reducing agent, or reductant, loses electrons and is oxidized in a chemical reaction. Examples of reducing agents include the earth metals, formic acid, and sulfite compounds. Definitions. A reducing agent reduces other substances and loses electrons; therefore, its oxidation state increases.Is NADH a reducing agent?
The coenzyme is, therefore, found in two forms in cells: NAD+ is an oxidizing agent – it accepts electrons from other molecules and becomes reduced. This reaction forms NADH, which can then be used as a reducing agent to donate electrons. These electron transfer reactions are the main function of NAD.What is the most powerful oxidizing agent?
fluorine
Is Oxygen an oxidizer?
No oxygen is not always an oxidizer or oxidising agent. For it to be so it needs to be reduced, I.e. It needs to gain electrons. For the most part oxygen almost always does this however, if it gets bonded to a much more electro negative atom such as fluorine then oxygen becomes oxidised and the fluorine reduced.Is sodium a reducing agent?
chemical compound classification reaction, sodium is called the reducing agent (it furnishes electrons), and chlorine is called the oxidizing agent (it consumes electrons). The most common reducing agents are metals, for they tend to lose electrons in their reactions with nonmetals.Is i2 a reducing agent?
Here, I(iodine) has seven electrons in its outermost shell and it tends to gain one electron to be as strong as the almighty nobel gases. It's just one electron away so it'll gain an electron and hence is an oxidizing agent. Reducing agent: substance which tends to reduce by getting oxidized and donating electrons.Is water an oxidizing agent?
In terms of redox, water behaves much as it did in acid-base reactions, where we found it to be amphiprotic. In the presence of a strong electron donor (strong reducing agent), water serves as an oxidizing agent. In the presence of a strong electron acceptor (strong oxidizing agent), water serves as a reducing agent.Is KMnO4 an oxidizing agent?
Potassium Permanganate ? Potassium permanganate is an inorganic chemical compound with the formula KMnO4. It is a salt consisting of K+ and MnO4 − ions. Formerly known as permanganate of potash or Condy's crystals, it is a strong oxidizing agent. It dissolves in water to give intensely purple solutions.Which metal is the strongest reducing agent?
Lithium
Is sodium an oxidizing agent?
Sodium is an alkali metal and so very easily oxidized to form Na+. So no, it is not an oxidizing agent. In fact, elemental sodium is a very good reducing agent and is widely used in inorganic and organic chemistry.What is mean by oxidizing agent?
An oxidizing agent is a reactant that removes electrons from other reactants during a redox reaction. The oxidizing agent typically takes these electrons for itself, thus gaining electrons and being reduced. An oxidizing agent is thus an electron acceptor. Oxidizing agents are also known as oxidants or oxidizers.Is carbon an oxidizing agent?
Each oxygen atom decreases its oxidation number from 0 to –2, so O2 is reduced and is the oxidizing agent. Thus carbon atoms in CO are both oxidized and reduced, and CO is both the oxidizing agent and the reducing agent.Which substance is the oxidizing agent in this reaction?
The oxygen atoms undergo reduction, gaining electrons, while the carbon atoms undergo oxidation, losing electrons. Thus oxygen is the oxidizing agent and carbon is the reducing agent in this reaction.What is oxidising agent give two examples?
Oxidizing agents are those which oxidizes the Other compound and removes hydrogen from the compound. Oxidizing agents are substances that gain electrons. Examples of oxidizing agents include halogens, potassium nitrate, and nitric acid.Is calcium a strong reducing agent?
The reducing agent is stronger when it has a more negative reduction potential and weaker when it has a more positive reduction potential. Common reducing agents include metals potassium, calcium, barium, sodium and magnesium, and also compounds that contain the H− ion, those being NaH, LiH, LiAlH4 and CaH2.Is h2so4 an oxidizing agent?
An oxidizing acid is a Brønsted acid that is a strong oxidizing agent. Again, copper has no reaction with dilute sulfuric acid, but in concentrated sulfuric acid, the highly acidic environment and high concentration of sulfate ions allow the sulfate ions to act as an oxidizing agent: Cu + 2 H2SO4>SO2+2H2O+SO4²- +Cu²+.Which is stronger oxidizing agent Hocl or hclo4?
Explanation: hocl is stronger oxidizing agent than hclo4 because it has the lowest oxidation state and hence it is a stronger oxidizing agent.Which is stronger reducing agent cu2+ or fe2+?
Answer: Cr2+ is a stronger reducing agent than Fe2+. This can be explained on the basis of the standard electrode potential values E°(Cr3+/Cr2+ = –0.41 V) and E° (Fe3+/Fe2+ = + 0.77 V). Thus Cr2+ is easily oxidised to Cr3+ but Fe2+ cannot be as readily oxidised to Fe3+.