Barfoed's reagent: Dissolve 13.3 g of copperacetate in 200 ml of distilled water and add 1.8 ml of glacialacetic acid to it. Method/Procedure: Take 1ml of testsample/solution in the tube. Add 2ml Barfoed'ssolution..
In this way, what is Barfoed's reagent?
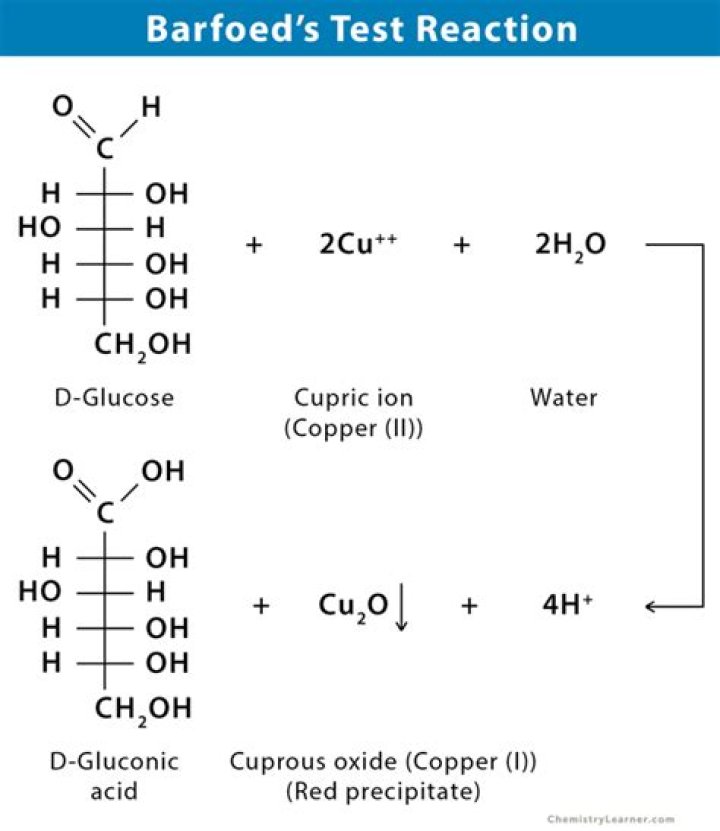
Barfoed's test is used to detect the presence ofmonosaccharide (reducing) sugars in solution. Barfoed'sreagent, a mixture of ethanoic (acetic) acid and copper(II)acetate, is combined with the test solution and boiled. A redcopper(II) oxide precipitate is formed will indicates the presenceof reducing sugar.
Also Know, how does Barfoed's test work? Principle : Barfoed's test reaction isbased on the reduction of cupric acetate by reducingmonosaccharides and reducing disaccharides. The reaction withdisaccharides is slower because disaccharides have to gethydrolyzed first and then react with the reagent cupric acetate toproduce cuprous oxide.
Considering this, what components make up the Barfoed's reagent?
Composition. Barfoed's reagent consists of a 0.33molar solution of neutral copper acetate in 1% acetic acidsolution. The reagent does not keep well and it is thereforeadvisable to make it up when it is actuallyrequired.
How do you make a Molisch reagent?
Dissolve the brown residue in minimum amount of NH3 to obtain a clear solution. (3)Molisch reagent:10% alcoholic solution of α-naphthol. (4)Barfoed'sreagent: to 100 cc of acetic acid in water, add 6.5 g ofcupric acetate. (5)Fehling's A: 17.3 g of CuSO4 in 250ml H2O containing few drops of conc.
Related Question Answers
What is Fehling solution A and B?
It is made initially as two separate solutions,known as Fehling's A and Fehling's B.Fehling's A is a blue aqueous solution of copper(II)sulfate pentahydrate crystals, while Fehling's B is a clearsolution of aqueous potassium sodium tartrate (also known asRochelle salt) and a strong alkali (commonly sodiumWhat is Seliwanoff's reagent?
Seliwanoff's test is a compound test whichseparates aldose and ketose sugars. Ketoses are differentiated fromaldoses by means of their ketone/aldehyde usefulness. If the sugarcontains a ketone bunch, it is a ketose. If the sugar contains analdehyde bunch, it is an aldose.Is Sucrose a disaccharide?
Sucrose is a disaccharide; each moleculeconsists of two "simple" sugars (a glucose and a fructose), calledmonosaccharides.How do you test for monosaccharides?
To test for the presence ofmonosaccharides and reducing disaccharide sugars in food,the food sample is dissolved in water, and a small amount ofBenedict's reagent is added.What is Seliwanoff's test used for?
Seliwanoff's test is a chemical test whichdistinguishes between aldose and ketose sugars. If the sugarcontains a ketone group, it is a ketose. If a sugar contains analdehyde group, it is an aldose. This test relies on theprinciple that, when heated, ketoses are more rapidly dehydratedthan aldoses.Is starch a monosaccharide?
Starch is a polysaccharide. Polysaccharides aresugars that contain more than one basic sugar unit.Monosaccharide, on the other hand, contain only one sugarmolecule. You can say that polysaccharides are polymers andmonosaccharides may become the monomers that build up thispolymer.What is the principle of Osazone test?
Osazones formation test involves the reaction ofa reducing sugar (free carbonyl group) with excess ofphenylhydrazine when kept at boiling temperature. All reducingsugars form osazones.Is maltose a reducing sugar?
The common dietary monosaccharides galactose, glucoseand fructose are all reducing sugars. Reducingdisaccharides like lactose and maltose have only one oftheir two anomeric carbons involved in the glycosidic bond, meaningthat they can convert to an open-chain form with an aldehydegroup.Is starch a disaccharide?
By definition a disaccharide is a carbohydratethat is formed of 2 monosaccharides. Starch is apolysaccharide, a much larger carbohydrate molecule, and iscomposed of long chains of many monosaccharides, (a great deal morethan 2).How are carbohydrates identified?
Carbohydrates can be represented by thestoichiometric formula (CH2O)n, where n is the number ofcarbons in the molecule. In other words, the ratio of carbon tohydrogen to oxygen is 1:2:1 in carbohydratemolecules.Is Lactose a disaccharide?
Lactose is a disaccharide. It is a sugarcomposed of galactose and glucose subunits and has the molecularformula C12H22O11. Lactosemakes up around 2–8% of milk (by weight). The name comes fromlac (gen. lactis), the Latin word for milk, plus the suffix -oseused to name sugars.Why is the Molisch test generally slower with the disaccharide sucrose?
Hence, it is able to distinguish monosaccharides fromdisaccharides. Monosaccharides are reducing sugars andtherefore, will react faster with the reagents to form the colourchange. Whereas, reducing disaccharides will reactslower than the monosaccharides.