Quick Answer. In a chemical equation, the location ofthe word "heat" can be used to quickly determine whether thereaction is endothermic or exothermic. If heat isreleased as a product of the reaction, the reaction isexothermic. If heat is listed on the side of thereactants, the reaction is endothermic..
Also, which is an exothermic process?
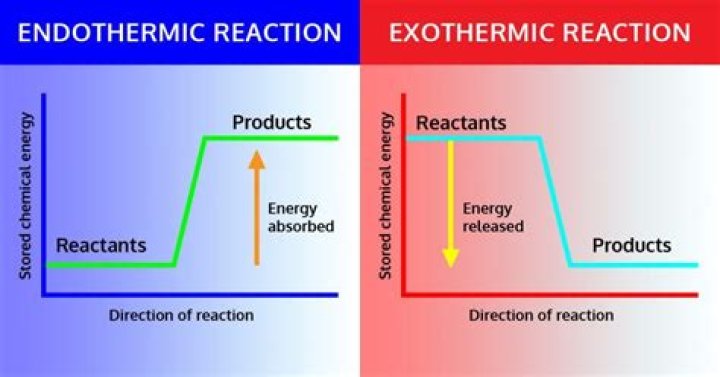
Exothermic- the word describes a processthat releases energy in the form of heat. Forming a chemical bondreleases energy and therefore is an exothermic process.Exothermic reactions usually feel hot because it is givingheat to you. Endothermic - a process orreaction that absorbs energy in the form ofheat.
Secondly, what is an example of an endothermic reaction? These examples could be written as chemicalreactions, but are more generally considered to beendothermic or heat-absorbing processes: Melting ice cubes.Melting solid salts. Converting frost to water vapor (melting,boiling, and evaporation, in general, are endothermicprocesses.
Similarly, what is the equation for exothermic reaction?
The general equation for an exothermicreaction is: Reactants → Products + Energy.
Why is freezing exothermic?
Exothermic by definition means the release ofheat from a system. Any process that slows down the particles inthe system due to heat flow outwards is thereforeexothermic. Freezing has particles of a liquid slowdown to form a lattice structure and become a solidphase.
Related Question Answers
Which is an endothermic process?
An endothermic process is any processwhich requires or absorbs energy from its surroundings, usually inthe form of heat. The opposite of an endothermic process isan exothermic process, one that releases, "gives out" energyin the form of heat.What happens in an exothermic process?
An exothermic reaction occurs when the energyused to break the bonds in the reactants (the starting stuff) isless than the energy released when new bonds are made in theproducts (the stuff you end up with). Combustion is an example ofan exothermic reaction- you can feel the heat given off ifyou get too close!Is water freezing endothermic?
Boiling is endothermic (it takes energy to boilwater), and so the water cools off as it boils.Eventually, what's left will freeze.Is cooking an egg endothermic or exothermic?
Endothermic absorbs heat, and exothermicproduces heat. Endothermic must be supplied with heat and isbasically the opposite of exothermic. An everyday reactionis in the cooking of an egg. There must be heat addedor absorbed from the environment to cook the egg orany other food item.Which is an exothermic reaction?
An exothermic reaction is a chemicalreaction that releases energy through light or heat. It isthe opposite of an endothermic reaction. Expressed in achemical equation: reactants → products + energy.Is water evaporating exothermic?
- Quora. An Exothermic Reaction will have anExothermic change, whereby energy (In the form of heat) isrelease/given out to the surrounding. Since Energy is Absorbed tobreak the bonds before it can become a gas ,that makesEvaporation an Endothermic Process.What happens in an endothermic reaction?
When energy is released as heat, the process isexothermic, and when heat is absorbed, the process isendothermic. An endothermic reaction is one whichresults in a net decrease in temperature because it absorbs heatfrom the surroundings and stores the energy in the bonds formed inthe reaction.Why is boiling water an endothermic process?
Boiling water is an endothermic process,which supplies heat to the water molecules, increasing theirpotential energy. The applied heat causes the watermolecules to move further away from each other without causing anyincrease in overall temperature.