How do you know if a compound is pure?
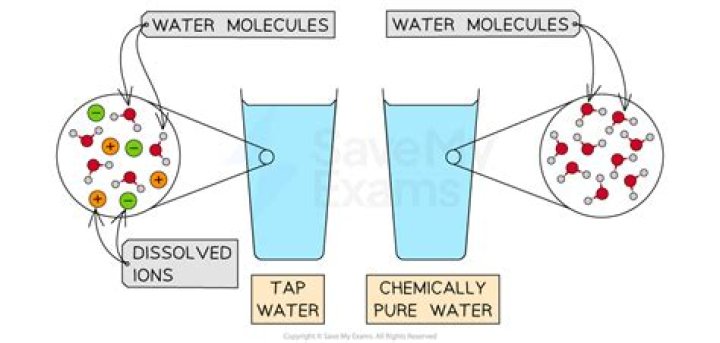
.
Also, what does a pure compound look like?
A pure compound is a compound, which is made up of two or more elements in the a particular ratio and that ratio never changes. For example, water contains hydrogen and oxygen in the ratio of 2:1. Additionally, when these elements are combined they have distinctive physical and chemical properties.
Similarly, what are pure liquids? In liquid: Behaviour of pure liquids. When the temperature and pressure of a pure substance are fixed, the equilibrium state of the substance is also fixed.
One may also ask, why do we need pure substances?
We need pure substances to: Measure accurate properties of the substance. Carry out chemical reactions without side effects of impurities. Use drugs for medical use without interference from impurities.
What is a pure compound?
Pure compounds are compounds consisting of one and only one type of molecule. Water, for example, is a pure compound because it consists only of H2O molecules. Gasoline is not a pure compound because it is a mixture of various hydrocarbons.
Related Question AnswersWhat are pure materials?
A pure substance or chemical substance is a material that has a constant composition (is homogeneous) and has consistent properties throughout the sample. In chemistry, a pure substance consists of only one type of atom, molecule, or compound.Is orange juice a pure substance?
A Orange juice contains particles of solid (pulp) as well as liquid; it is not chemically pure. B Because its composition is not uniform throughout, orange juice is a heterogeneous mixture. A Aluminum oxide is a single, chemically pure compound. A Selenium is one of the known elements.What are the types of substances?
Types of Substances- Pure Substance: The substances that are free from any kind of mixture and contain only one kind of particle are pure substances. Examples of pure substances include iron, aluminum, silver, and gold.
- Mixtures: Substances that have two or more different particles are mixtures.
What are the two types of pure substances?
There are two types of pure substances that are Elements and Compounds. Examples of elements are: Iron, Silver, Gold, Mercury etc. Examples of compounds are: Water, Carbon dioxide, methane, vinegar etc.What is a example of a pure substance?
Examples of pure substances include tin, sulfur, diamond, water, pure sugar (sucrose), table salt (sodium chloride) and baking soda (sodium bicarbonate). Crystals, in general, are pure substances. Tin, sulfur, and diamond are examples of pure substances that are chemical elements. All elements are pure substances.What is an example of a compound?
A compound is a substance that is made up of two or more elements. Some examples of compounds include the following: water, carbon dioxide, and table salt.What does a compound look like?
Compound: two or more different atoms bonded together. Mixture: two or more different atoms together but not joined. Molecule: two particles (same or different) bonded together. Element: only 1 type of atom; this definition is applied to things both bonded and not to itself.How do you know how many elements are in a compound?
To find the molar mass of a compound:- Use the chemical formula to determine the number of each type of atom present in the compound.
- Multiply the atomic weight (from the periodic table) of each element by the number of atoms of that element present in the compound.