How do you find density of a gas?
.
Subsequently, one may also ask, what is the density of a gas?
Gas density is defined to be the mass of gas divided by the volume confining the gas. There is a related state variable called the specific volume which is the reciprocal of the density r. The specific volume v is given by: v = 1 /r.
Also, what is the formula for density of a gas? To find density, we have to solve the equation for volume, or V. V = nRT / P. To incorporate mass, we can use the number of moles, or n. The number of moles equals the mass of the gas divided by the molecular mass.
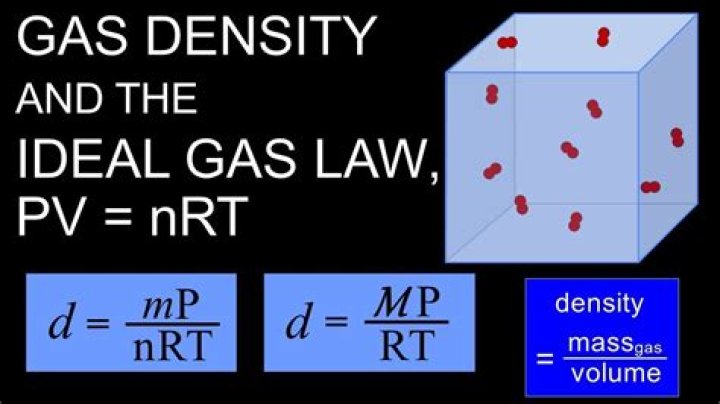
Moreover, how do you find density using the ideal gas law?
The original ideal gas law uses the formula PV = nRT, the density version of the ideal gas law is PM = dRT, where P is pressure measured in atmospheres (atm), T is temperature measured in kelvin (K), R is the ideal gas law constant 0.0821 atm(L)mol(K) just as in the original formula, but M is now the molar mass ( gmol
What is specific gravity of gas?
Specific gravity. Apparent specific gravity is the ratio of the weight of a volume of the substance to the weight of an equal volume of the reference substance. The reference substance for liquids is nearly always water at its densest (at 4 °C or 39.2 °F); for gases it is air at room temperature (20 °C or 68 °F).
Related Question AnswersIs density a weight?
Weight density is weight of a substance per unit volume. Our definitions of mass and weight are made in such a way that weight in Kg is same as mass in Kg on earth surface in standard reference place. So an object will have same mass on earth and moon, but weight will be different at these two places.How do you measure gas?
Use a gas syringe - This is a laboratory instrument that can be used to withdraw a certain volume of gas from a closed system or to measure the volume of gas from a chemical reaction. Use a spirometer - It measures the volume of air coming into and being expelled out of your lungs, called tidal volume.Which gas has highest density?
The ultimate gas density would be uranium hexachloride using depleted uranium and Cl-37, with a molar mass of 460 g/mol, which makes it over 50 % denser in gas form than tungsten hexafluoride, and 3 times denser than sulfur hexafluoride, but it has a boiling point of 75 ∘C, decomposes on contact with air, is toxic andWhy is the density of gas low?
Gases have lower density compared to that of solids or liquids. Explain. Gases are less dense than solids and liquids because their molecules are far apart in comparison to solids and liquids. Because the molecules of a gas are spread apart, they have the least volumemass, and are therefore less dense.Does gas have high density?
A gas usually has much lower density than a solid or liquid. The molecules or atoms in a gas are much further apart than in a solid or a liquid. Gas molecules or atoms are usually flying around at very high speeds, occasionally bouncing off each other or the walls of the container the gas is in.What is mass of gas?
The molecular weight (molar mass) of any gas is the mass of one particle of that gas multiplied by Avogadro's number (6.02 x 1023). Knowing the molar mass of an element or compound can help us stoichiometrically balance a reaction equation.What unit is density measured in?
The formula for density is d = M/V, where d is density, M is mass, and V is volume. Density is commonly expressed in units of grams per cubic centimetre. For example, the density of water is 1 gram per cubic centimetre, and Earth's density is 5.51 grams per cubic centimetre.What is the density of n2o a gas at STP?
1.22 g / cm3What is the density of solid?
Density is a measure of how compact the mass in a substance or object is. Divide the mass by the volume to figure out the density (p = m / v). You may also need to know how to calculate the volume of a solid. For example, for a cube, volume is length times width times height (V = L * W * H).What is PV nRT called?
PV = nRT: The Ideal Gas Law. Fifteen Examples Each unit occurs three times and the cube root yields L-atm / mol-K, the correct units for R when used in a gas law context. Consequently, we have: PV / nT = R. or, more commonly: PV = nRT. R is called the gas constant.What is the equation for volume?
The formula to find the volume multiplies the length by the width by the height. The good news for a cube is that the measure of each of these dimensions is exactly the same. Therefore, you can multiply the length of any side three times. This results in the formula: Volume = side * side * side.What is the value of R?
The value of the gas constant 'R' depends on the units used for pressure, volume and temperature. R = 0.0821 liter·atm/mol·K. R = 8.3145 J/mol·K. R = 8.2057 m3·atm/mol·K. R = 62.3637 L·Torr/mol·K or L·mmHg/mol·K.What is the density of so2 gas at STP?
| Gas | Density (g/L) |
|---|---|
| hydrogen chloride | 1.639 |
| methane | 0.7165 |
| sulphur dioxide | 2.927 |
| nitrogen | 0.716 |



