How do you determine R or S configuration?
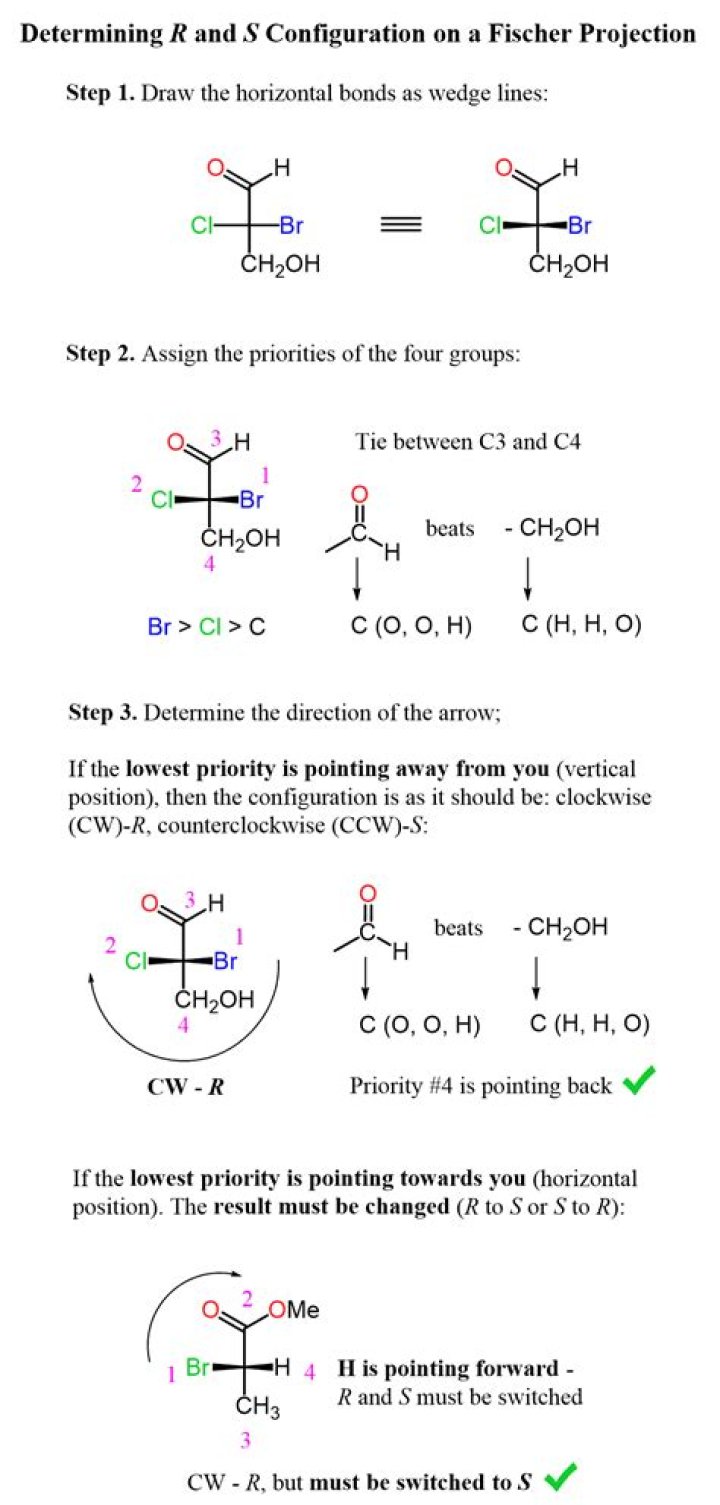
.
Consequently, how do you know if a chiral center is R or S?
If a molecule has a chiral center that is designated R, the chiral center will be S in the molecule's enantiomer. You need to be able to assign whether a chiral center is R or S. To do so, you need to follow three steps: Number each of the substituents on the chiral center carbon using the Cahn–Ingold–Prelog system.
Subsequently, question is, what does R and S mean in stereochemistry? The Cahn-Ingold-Prelog system is a set of rules that allows us to unambiguously define the stereochemical configuration of any stereocenter, using the designations 'R ' (from the Latin rectus, meaning right-handed) or ' S ' (from the Latin sinister, meaning left-handed).
Secondly, which molecule has the R configuration?
The circle is clockwise, which by step 4a tells us that this carbon has the 'R' configuration, and that this molecule is (R)-glyceraldehyde. Its enantiomer, by definition, must be (S)-glyceraldehyde.
Are R and S enantiomers?
Stereocenters are labeled R or S The "right hand" and "left hand" nomenclature is used to name the enantiomers of a chiral compound. The stereocenters are labeled as R or S. Consider the first picture: a curved arrow is drawn from the highest priority (1) substituent to the lowest priority (4) substituent.
Related Question AnswersWhat is R configuration?
R indicates that a clockwise circular arrow that goes from higher priority to lower priority crosses over the lowest priority substituent and that lowest-priority substituent is in the back.How do I assign priority to R and S?
Assign the priority (high = 1 to low = 4) to each group attached to the chirality center based on atomic number. Reposition the molecule so that the lowest priority group is away from you as if you were looking along the C-(4) σ bond. If you are using a model, grasp the lowest priority group in your fist.What does it mean to be chiral?
Definitions: Chiral. A molecule is chiral if it is not superimposable on its mirror image. Most chiral molecules can be identified by their lack of a plane of symmetry or a center of symmetry. Your hand is a chiral object, as it does not have either of these types of symmetry.How do you determine chirality?
We can look for a plane of symmetry in the molecule. Imagine this plane as a mirror through the middle of the molecule. If one half of the molecule is reflected into the other half, then the molecule is achiral. If no such mirror plane exist, the molecule is usually chiral.What is D and L configuration?
d- and l- is the exact same with (+) and (-) notation. Additional explanation. D-L system (also called Fischer–Rosanoff convention) is mainly used for naming α-amino acids and sugars. It compares the relative configurations of molecules to the enantiomers of glyceraldehyde. This convention is still in common use today.What is R and S notation?
R and S Notation[edit] Follow the direction of the remaining 3 priorities from highest to lowest priority (lowest to highest number, 1<2<3). A counterclockwise direction is an S (sinister, Latin for left) configuration. A clockwise direction is an R (rectus, Latin for right) configuration.What is the difference between enantiomers and diastereomers?
There are two types of stereoisomers— enantiomers and diastereomers. Enantiomers contain chiral centers that are mirror images and non-superimposable. Diastereomers contain chiral centers that are non-superimposable but are NOT mirror images. There can be many more than 2 depending on the number of stereocenters.How do you assign a chiral center?
There are two steps to assigning a chiral center.- Start with the first atom of each group that is directly attached to the chiral tetrahedral center.
- If two groups have the same first atom, then compare the second atom from the chiral center.



