How do you determine R and S configuration in Fischer projection?
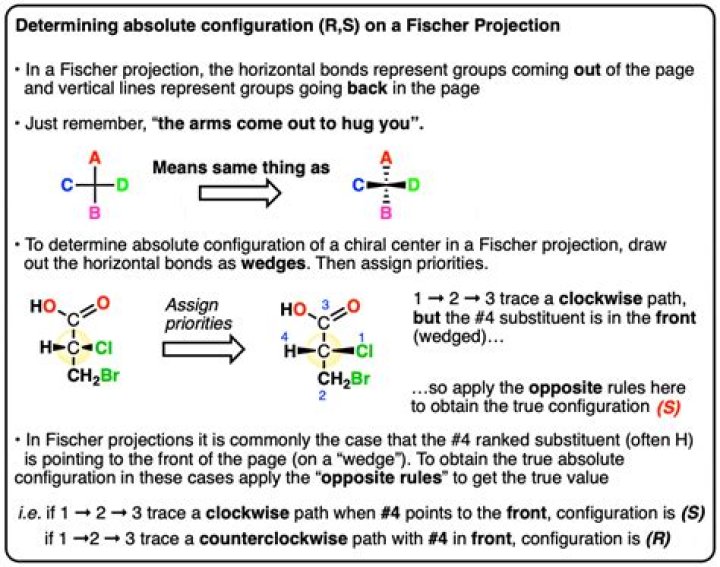
.
Hereof, how do I assign priority to R and S?
Assign the priority (high = 1 to low = 4) to each group attached to the chirality center based on atomic number. Reposition the molecule so that the lowest priority group is away from you as if you were looking along the C-(4) σ bond. If you are using a model, grasp the lowest priority group in your fist.
what is Haworth projection formula? A Haworth projection is a common way of writing a structural formula to represent the cyclic structure of monosaccharides with a simple three-dimensional perspective. Organic chemistry and especially biochemistry are the areas of chemistry that use the Haworth projection the most.
Beside this, what is the difference between enantiomers and diastereomers?
There are two types of stereoisomers— enantiomers and diastereomers. Enantiomers contain chiral centers that are mirror images and non-superimposable. Diastereomers contain chiral centers that are non-superimposable but are NOT mirror images. There can be many more than 2 depending on the number of stereocenters.
What is sawhorse formula?
A saw-horse formula is a diagram used to depict a specific conformation of a molecule. eg: The staggered conformation of ethane is.
Related Question AnswersWhat is Fischer projection formula?
A Fischer projection or Fischer projection formula is a convention used to depict a stereoformula in two dimension without destroying the stereochemical information, i.e., absolute configuration, at chiral centers.Are hands chiral or achiral?
When the thumbs point in the same direction, the palms are opposite. Your hands are mirror images, but not superposable. Each hand is therefore chiral. Achiral objects may be superposed on their mirror image.What is meant by R and S configuration?
The Cahn-Ingold-Prelog system is a set of rules that allows us to unambiguously define the stereochemical configuration of any stereocenter, using the designations 'R ' (from the Latin rectus, meaning right-handed) or ' S ' (from the Latin sinister, meaning left-handed).How do you know if a chiral center is R or S?
Draw a curve from the first-priority substituent through the second-priority substituent and then through the third. If the curve goes clockwise, the chiral center is designated R; if the curve goes counterclockwise, the chiral center is designated S.What does it mean to be chiral?
Definitions: Chiral. A molecule is chiral if it is not superimposable on its mirror image. Most chiral molecules can be identified by their lack of a plane of symmetry or a center of symmetry. Your hand is a chiral object, as it does not have either of these types of symmetry.Which functional group has highest priority?
According to IUPAC convention, Carboxylic Acids and their derivatives have the highest priority then carbonyls then alcohols, amines, alkenes, alkynes, and alkanes, so in this case the Carboxylic acid group has the highest priority and therefore makes up the name of the base compound.How many stereoisomers are possible for?
Three stereoisomers are possible: one pair of enantiomers (A and B) and an achiral molecule C, called a “meso compound.” A meso compound is an achiral molecule that nonetheless contains a stereogenic atom.What is D and L configuration?
d- and l- is the exact same with (+) and (-) notation. Additional explanation. D-L system (also called Fischer–Rosanoff convention) is mainly used for naming α-amino acids and sugars. It compares the relative configurations of molecules to the enantiomers of glyceraldehyde. This convention is still in common use today.What is R and S enantiomers?
The R / S system is an important nomenclature system for denoting enantiomers. This approach labels each chiral center R or S according to a system by which its substituents are each assigned a priority, according to the Cahn–Ingold–Prelog priority rules (CIP), based on atomic number.What is D in organic chemistry?
Deuterium is frequently represented by the chemical symbol D. Since it is an isotope of hydrogen with mass number 2, it is also represented by 2. H. .What is the highest priority substituent group?
The highest priority group has the highest priority atom. In this case we compare HHH on the CH3 versus CHH on the ethyl. Hydrogen is lower atomic number and therefore lower priority than carbon, so the methyl group has lower priority than the ethyl group.How do I configure D and L configuration?
Step 1: Make sure that the molecule is drawn as the Fischer projection in which the carboxylic acid group is on top and the side chain on bottom. eg: Step 2: If the amine group is on the right of the carbon chain, assign the label D to the compound; if it is on the left of the carbon chain, assign the label L.Are all meso compounds identical?
Meso compounds are therefore non-chiral and optically inactive. "Meso" comes from the Greek for "middle" (relating to the presence of the plane of symmetry that divides the structure down the middle). It is not specifically a synonym for "identical".What do horizontal and vertical positions mean in Fischer projection?
The Fischer Projection consists of both horizontal and vertical lines, where the horizontal lines represent the atoms that are pointed toward the viewer while the vertical line represents atoms that are pointed away from the viewer.How do you choose R and S configuration?
After all your substituents have been prioritized in the correct manner, you can now name/label the molecule R or S.- Put the lowest priority substituent in the back (dashed line).
- Proceed from 1 to 2 to 3. (
- Determine if the direction from 1 to 2 to 3 clockwise or counterclockwise.



