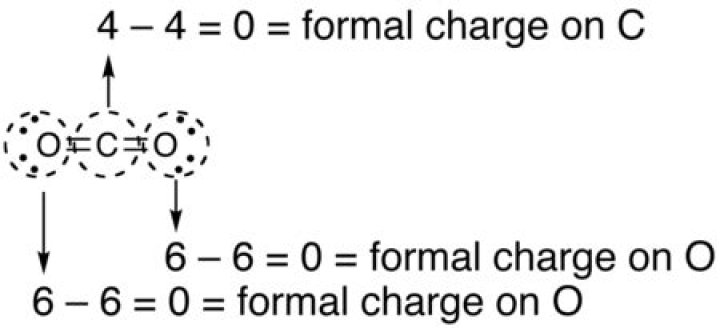Determining Formal Charge In this formula, V represents the number of valence electrons of the atom in isolation, N is the number of non-bonding valence electrons, and B is the total number of electrons in covalent bonds with other atoms in the molecule. The oxygen atom in carbon dioxide has a formal charge of 0..
Simply so, what is the charge for co2?
Since carbon has 4 valence electrons, its formal charge will be zero. The same is true for both oxygen atoms. Both of them form 2 bonds, which means they get 2 electrons. In addition to these electrons, they both have 2 lone pairs; this brings the total number of electrons an oxygen atom gets to 6 (2 + 4).
Additionally, does co2 have a positive formal charge? In carbon dioxide, the carbon atom has double bonds to oxygen on both sides (O=C=O). Later on in this chapter and throughout this book we will see examples of organic ions called 'carbocations' and carbanions', in which a carbon atom bears a positive or negative formal charge, respectively.
Regarding this, what is the formal charge on carbon?
Carbocations occur when a carbon has only three bonds and no lone pairs of electrons. Carbocations have only 6 valence electrons and a formal charge of +1. Carbanions occur when the carbon atom has three bonds plus one lone pair of electrons. Carbanions have 8 valence electrons and a formal charge of -1.
What is the oxidation number of co2?
The oxidation number of C in carbon dioxide (CO2) is (rules 1 & 2): 0 + (2 x 2) = +4 [Check (rule 3): +4 + 2(-2) = 0] The oxidation number of C in methane (CH4) is (rules 1 & 2): 0 – (4 x1) = -4 [Check (rule 3): -4 + 4(-1) = 0].
Related Question Answers
What is the charge of water?
The unequal sharing of electrons gives the water molecule a slight negative charge near its oxygen atom and a slight positive charge near its hydrogen atoms. When a neutral molecule has a positive area at one end and a negative area at the other, it is a polar molecule.How do you determine hybridization?
A Shortcut For Determining The Hybridization Of An Atom In A Molecule - Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.
How many nonbonding electrons are in co2?
When the nonbonding electrons are used to satisfy the octet of the oxygen atom, the carbon atom has a total of only six valence electrons. We therefore assume that the carbon and oxygen atoms share two pairs of electrons. There are now four bonds in the skeleton structure, which leaves only four nonbonding electrons.Is co2 polar or nonpolar?
Carbon dioxide is non-polar because of the symmetry of its bonding. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are at 180 degrees to each other so the dipoles cancel out.What is a co2 cleaner?
Carbon dioxide cleaning (CO2 cleaning) comprises a family of methods for parts cleaning and sterilization, using carbon dioxide in its various phases. It is often preferred for use on delicate surfaces. CO2 cleaning has found application in the aerospace, automotive, electronics, medical, and other industries.What is formal charge formula?
However, since the “number of bonding electrons divided by 2” term is also equal to the number of bonds surrounding the atom, here's the shortcut formula: Formal Charge = [# of valence electrons on atom] – [non-bonded electrons + number of bonds]. The number of non-bonded electrons is zero.What is formal charge?
In chemistry, a formal charge (FC) is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity.Which formal charge is best?
Our general rule is that the best structure minimizes the formal charges. This is because minimizing the formal charges leads to the electrons being most evenly distributed about the different atomic centers in a molecule. Having electrons concentrated in one area will lead to regions of negative charge.What is formal charge Class 11?
A Formal charge is also known as a Fake Charge. It's a theoretical charge over an individual atom of an ion as the real charge over a polyatomic molecule or ion is distributed on an ion as a whole and not over a single atom. The formal charge of an atom of a polyatomic molecule or ion is defined below.What is the formal charge of oxygen in h2o?
The formal charge on oxygen is calculated as follows. Oxygen has six valence electrons (GN = 6), two unshared electrons in one lone pair (UE = 2), and six shared electrons in three oxygen-hydrogen covalent bonds (BE = 6). Thus the calculated formal charge on oxygen is +1.How can formal charges be reduced?
If a formal charge of 1- is located next to a formal charge of 1+, the formal charges can usually be minimized by having a lone pair of electrons, located on the atom with the 1- charge become a bonding pair of electrons that is shared with the atom that has the 1+ formal charge (this can be visualised in the same wayDoes co2 have a resonance structure?
Carbon dioxide, or CO2, has three resonance structures, out of which one is a major contributor. The CO2 molecule has a total of 16 valence electrons , 4 from carbon and 6 from each oxygen atom. Structures 2 and 3 show charge separation caused by the presence of formal charges on both oxygen atoms.Does h2o have charge?
Water, H2O, is a polar molecule, the partial negative charge being placed on the oxygen atom and the positive partial charge distributed on the two hydrogen atoms. The H+ ion or proton, is hydrated and can be written H3O+ or H9O4+ depending whether the positive charge H+ is distributed on one or four water molecules.Why do we calculate formal charge?
Knowing the formal charge on a particular atom in a structure is an important part of keeping track of the electrons and is important for establishing and predicting the reactivity. The formal charge on an atom in a molecule reflects the electron count associated with the atom compared to the isolated neutral atom.Does carbon have a positive or negative charge?
Neutral carbon-12 (or any carbon atom) has 6 electrons with a total negative charge of 6e- orbiting a nucleus with a total positive charge of 6e+, so that the total net charge is zero. The nucleus is made up of 6 protons, each with a positive charge of e+, and 6 neutrons, each with zero charge.Is oxygen positive or negative?
A good example is an oxygen atom that is sharing one electron with each of two nearby hydrogen atoms, making a molecule of water H2O. An oxygen atom that has successfully donated one or two electrons or is sharing one or two electrons has lost part of its negative charge, so it is left positively charged.What is the oxidation state of CO?
In its compounds cobalt nearly always exhibits a +2 or +3 oxidation state, although states of +4, +1, 0, and −1 are known.Why is bh4 negatively charged?
And the Boron has 8 valence electrons. So we've used all 8 valence electrons for the BH4 Lewis structure, and each of the atoms has a full outer shell. One last thing we need to do is put brackets around the ion to show that it has a negative charge. So that's the Lewis structure for BH4-, the tetrahydroborate ion.Is hydrogen positive or negative?
One Hydrogen (H) has a positive charge +1. This has nothing to do with it having more protons than electrons, as it has one of each. Think of it as 'willing' to donate one electron, or it has +1 electrons see, it is on the plus side. One Oxygen (O) has a negative charge of -2.