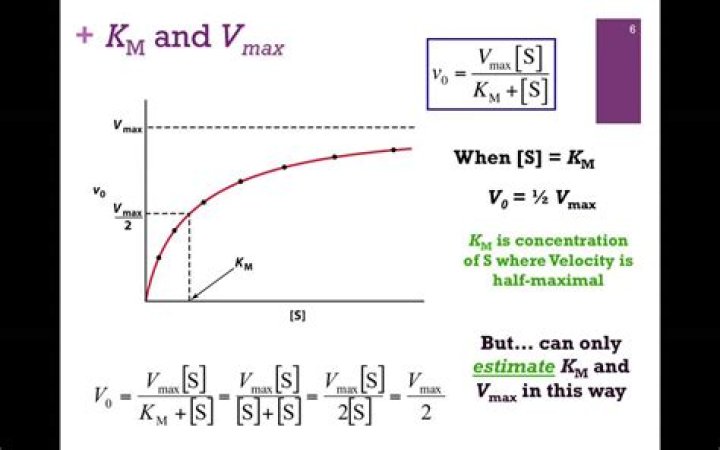
Km and Vmax are determined by incubating the enzyme with varying concentrations of substrate; the results can be plotted as a graph of rate of reaction (v) against concentration of substrate ([S], and will normally yield a hyperbolic curve, as shown in the graphs above..
Consequently, how do you calculate km?
You can estimate KM and Vmax from the graph of initial velocity versus [S].
- Run a series of reactions with constant [Etot], varying [S], and measure Vo.
- Graph Vo vs. [S].
- Estimate Vmax from asymptote.
- Calculate Vmax/2.
- read KM from graph.
Also, what are the units of Km and Vmax? The units of Km are those of concentration i.e. mM, mM or Km is the concentration of substrate at which half maximal velocity is observed. Vmax can be expressed in a variety of units depending on what information is available.
Considering this, what is the unit of Vmax?
Vmax "represents the maximum rate achieved by the system, at maximum (saturating) substrate concentrations" (wikipedia). Unit: umol/min (or mol/s). But then the enzymatic activity of a sample is the amount of enzyme that converts 1 umole of substrate/min in the optimal conditions
What is Km value?
The Michaelis constant (KM) is defined as the substrate concentration at which the reaction rate is half of its maximal value (or in other words it defines the substrate concentration at which half of the active sites are occupied).
Related Question Answers
Can km be negative?
If you follow the disappearance of something, the velocity should be "negative" and hence you need to invert it to get a positive reaction velocity. Only when the reaction rate is positive will you find both Michaelis-Menten parameters to be positive. Also, make sure your reaction rate is faster as [S] increases.What are the units of kcat?
The unit of Kcat is in 1/sec. The reciprocal of Kcat is then the time required by an enzyme to "turn over" a substrate molecule. The higher the Kcat is, the more substrates get turned over in one second. Km is the concentration of substrates when the reaction reaches half of Vmax.Is kcat the same as Vmax?
Kcat is equal to Vmax/[Enzyme]. Because the concentration of enzyme is taken into account in this equation, Kcat does NOT vary with the amount of enzyme used and is therefore a constant for an enzyme. Kcat is equal to the number of molecules of product made per enzyme per unit time.What is Lineweaver Burk equation?
The Lineweaver-Burk equation is a linear equation, where 1/V is a linear function of 1/[S] instead of V being a rational function of [S]. The Lineweaver-Burk equation can be readily represented graphically to determine the values of Km and Vmax.What is a high Vmax value?
The rate of reaction when the enzyme is saturated with substrate is the maximum rate of reaction, Vmax. This is usually expressed as the Km (Michaelis constant) of the enzyme, an inverse measure of affinity. For practical purposes, Km is the concentration of substrate which permits the enzyme to achieve half Vmax.What does kcat mean?
Kcat is the turnover number -- the number of substrate molecule each enzyme site converts to product per unit time. If you know the concentration of enzyme sites, you can fit Kcat instead of Vmax when analyzing a substrate vs. velocity curve.How do you make a Michaelis Menten graph?
Using graph paper, draw an x- and y-axis. Label the x-axis mM of [S] or concentration of substrate. Label the y ax- sec/micro-mole of V or velocity of reaction. Insert different values of [S] into the Michaelis-Menten equation, along with the values found for Km and Vmax, to solve for V.How does Michaelis Menten calculate km?
The equation that defines the Michaelis-Menten plot is: V = (Vmax [S]) ÷ (KM + [S}). At the point at which KM = [S], this equation reduces to V = Vmax ÷ 2, so KM is equal to the concentration of the substrate when the velocity is half its maximum value.What is kcat km?
Kcat/Km represents the rate of the reaction at negligible substrate concentration. Or in other words, Kcat/Km is the (pseudo-)second order rate constant between the enzyme and the substrate, when [S]≪Km[S]≪Km.This still leaves the issue of why Kcat/Km is often referred to as the "specificity constant" of the enzyme.Which enzyme has the highest catalytic efficiency?
ELB17 displays the highest catalytic efficiency of all HLDs found in the marine environment with kcat/KM = 88.1 s−1 mM−1 with 1,3-dibromopropane (Chrast et al., 2018).What is Vmax physics?
vmax = maximum velocity at equilibrium (m/s) A = amplitude of mass (m) k = spring constant (N/m) m = mass (kg) Example 2: A 17kg mass is pulled 13cm away from its equilibrium point, on a spring with a 367 N/m constant.Is km a distance?
Distance Unit: is the unit of distance, you can choose kilometers, miles or meters. Kilometers (km): is the unit of length equal to 1000 meters or 0.62137 miles.What does a low km mean?
4. Since the Michaelis-Menton constant Km is the concentration of substrate at 0.5Vmax, it is an inverse measure of its substrate affinity, because a lower Km indicates that less substrate is needed to reach a certain reaction speed. Hence, a low Km means a high substrate affinity.What is the slope of a Lineweaver Burk plot?
The Lineweaver-Burk plot results in a straight line with the slope equal to KM/k2[E]0 and y-intercept equal to 1/k2[E]0 which is 1/Vmax via Equation 10.2.How do you calculate the velocity of a reaction?
The reaction velocity (v) equals (Vmax [A])/(Km + [A]) as described by the Michaelis-Menten equation where Vmax is the maximal velocity, [A] is the substrate concentration, and Km is the Michaelis constant, or the substrate concentration at half maximal velocity.Why are Lineweaver Burk plots inaccurate?
The Lineweaver–Burk plot is classically used in older texts, but is prone to error, as the y-axis takes the reciprocal of the rate of reaction – in turn increasing any small errors in measurement. Also, most points on the plot are found far to the right of the y-axis.How do you calculate kcat?
Note: the enzyme concentration is the same for all of the test tubes; only the substrate concentrations vary in the assay. Divide the Vmax (from Section 2, Step 4) by the enzyme concentration (from Section 2, Step 5). The result is the value of Kcat.How do you calculate km per hour?
Convert Miles per Hour to Kilometers per Hour Convert the speed from miles per hour to kilometers per hour. If the speed is given in miles per hour, then multiply by 1.609. If the speed is measured in meters per second (m/s), multiply by 3.6.Why is Vmax never reached?
Why is the Vmax of an enzyme an asymptotic non-reachable value? The thermodynamics of binding tells us that the active site can never reach 100% bound, and thus the enzyme can never achieve a reaction rate of Vmax, but as substrate is increased for an ideal enzyme the reaction rate approaches Vmax.