From Simple to Complex Super-tiny subatomic particles are used to create the parts of atoms. Protons, neutrons, and electrons can then organize to form atoms. Atoms are then used to create the molecules around us. As we just learned, there are almost 120 elements that can be found in the molecules we know..
Keeping this in view, how do atoms make up matter?
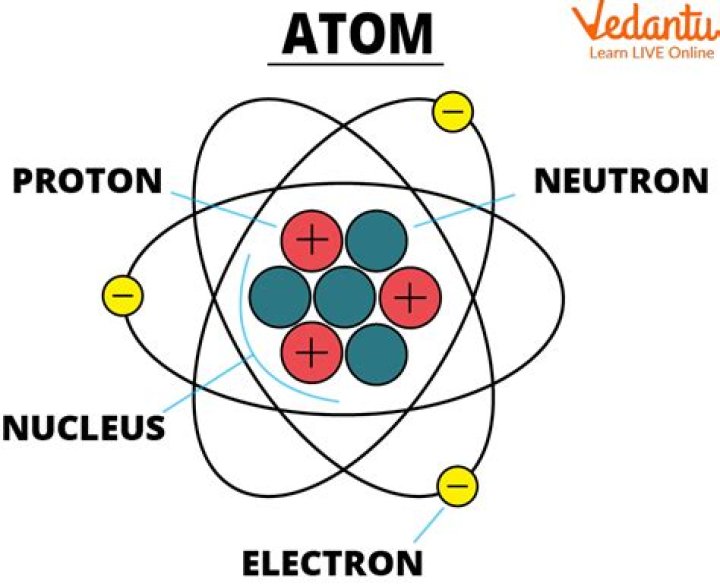
Atoms are the basic building blocks of ordinary matter. Atoms can join together to form molecules, which in turn form most of the objects around you. Atoms are composed of particles called protons, electrons and neutrons.
do atoms make up elements? Elements. An element is a substance made up of atoms with the same number of protons. Elements are the simplest substances known. They can be metals (e.g. iron, copper, sodium magnesium) or non-metals (e.g. carbon, hydrogen, oxygen, nitrogen).
Keeping this in consideration, what are atoms and elements?
) A particular atom will have the same number of protons and electrons and most atoms have at least as many neutrons as protons. An element is a substance that is made entirely from one type of atom. For example, the element hydrogen is made from atoms containing just one proton and one electron.
Why elements are called the building blocks of matter?
Because all matter is composed of one element or a combination of two or more elements. A pure substance made of two or more elements, chemically combined & in a specific ratio.
Related Question Answers
Is there anything not made of atoms?
Neutron stars are made out of neutrons, so there's definitely no atoms there. The sparse matter which is between stars or between galaxies, which is the majority of the mass in the universe (!) (not including dark matter), is also not mostly made of atoms. It's apparently a plasma-like mix of protons and electrons.Can you see an atom?
Atoms are so small that we cannot see them with our eyes (i.e., microscopic). To give you a feel for some sizes, these are approximate diameters of various atoms and particles: atom = 1 x 10-10 meters. neutron or proton = 1 x 10-15 meters.What are the 4 types of atoms?
Different Kinds of Atoms - Description. Atoms are made of tiny particles called protons, neutrons and electrons.
- Stable. Most atoms are stable.
- Isotopes. Every atom is a chemical element, like hydrogen, iron or chlorine.
- Radioactive. Some atoms have too many neutrons in the nucleus, which makes them unstable.
- Ions.
- Antimatter.
How many atoms are in the human body?
In summary, for a typical human of 70 kg, there are almost 7*1027 atoms (that's a 7 followed by 27 zeros!) Another way of saying this is "seven billion billion billion." Of this, almost 2/3 is hydrogen, 1/4 is oxygen, and about 1/10 is carbon. These three atoms add up to 99% of the total!Can atoms be destroyed?
Atoms cannot be created nor destroyed, and they are indestructible; they cannot be broken into smaller parts. This was based on the Law of Conservation of Mass. It was later learned that atoms can break into smaller parts. When atoms of different elements form compounds, they combine in a simple, whole number ratio.Do atoms die?
Atoms are the building blocks of matter. They, quite literally, make our universe what it is. When we die, our bodies do not turn into nothing; rather, they are broken down into their constituent parts and recycled into the ecosystem. In short, our atoms go on long after we are gone.Are atoms living or nonliving?
If atoms were alive, than everything (except energy) would be alive because all matter is made of atoms. Atom is non living and will continue to be in its physical state. The live cells breathe and the atoms just continue to be and are not alive.How small is an atom?
You probably already know everything is made up of little tiny things called atoms or even that each atom is made up of even smaller particles called protons, neutrons and electrons. And you've probably heard that atoms are small.Can we create an atom?
Theoretically, the universe created atoms by its nature as it cooled. And let's not forget that an atom is a complex combination of elementary particles, so before we could create an atom, we'd need to master creating, at the very least, protons and electrons, and before them, quarks and gluons.Why are atoms so important?
Atoms are extremely important structures that make up all of the materials on earth. Atoms are in our bodies and they bond together to form molecules, which make up matter.Is an element smaller than an atom?
An element is a substance completely made up of one atom. However, the atom itself is not the smallest known particle, but instead each atom is made up of three individual parts: electrons, protons and neutrons. Furthermore, protons and neutrons themselves are made up of even smaller parts called quarks.Can an element be a molecule?
A molecule is the smallest particle of a substance that exists independently. Molecules of most elements are made up of only one of atom of that element. Oxygen, along with nitrogen, hydrogen, and chlorine are made up of two atoms. A compound is a substance formed when two or more elements are chemically joined.Who discovered atom?
Democritus
Which is larger atom or element?
An element is more the “subject” such as sodium, carbon, iron etc are elements. An element can be a single gold atom or a whole chuck of pure gold (Au). An atom is the single distinguishing component which makes an element. For example a single carbon (name of the element) atom has 6 protons and 6 electrons.What is the simplest element?
Hydrogen is the simplest element; an atom consists of only one proton and one electron. It is also the most plentiful element in the universe.What is an atom in chemistry?
An atom is the smallest unit of matter that has the properties of an element. It is composed of a dense core called the nucleus and a series of outer shells occupied by orbiting electrons. The nucleus, composed of protons and neutrons, is at the center of an atom.How many natural elements are there?
Of these 118 elements, 94 occur naturally on Earth. Six of these occur in extreme trace quantities: technetium, atomic number 43; promethium, number 61; astatine, number 85; francium, number 87; neptunium, number 93; and plutonium, number 94.What is bigger than a molecule?
A uranium atom has about 239 times as much mass as a hydrogen atom. This means that a big atom can be more massive than a molecule that is made up of multiple smaller atoms. And of course, a small atom can be less massive than a molecule.What is the number of atoms in an element?
To calculate the number of atoms in a sample, you need to find how many moles of the element the sample contains. A mole is a unit chemists use. It's equal to Avogadro's number (6.02 X 1023) of atoms. By definition, the weight of one mole of an element (its molar mass) is equal to its atomic weight in grams.