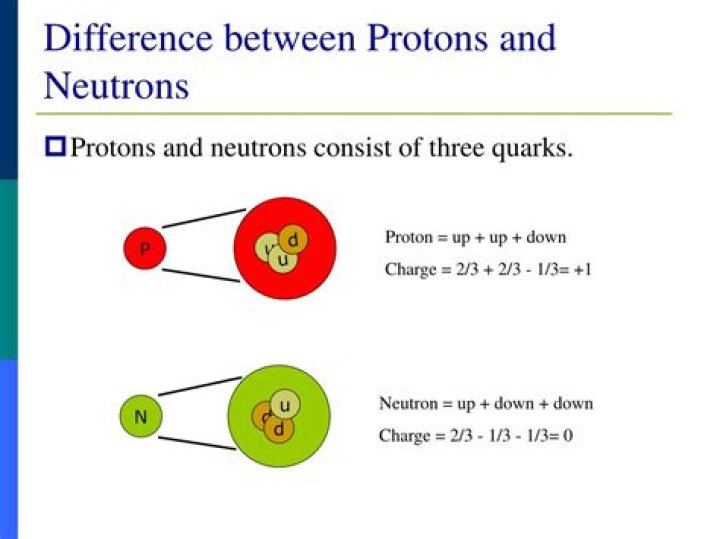
How are protons and neutrons similar and different?
.
Likewise, how are protons and electrons similar and different?
Electrons and protons are similar in that both are charged sub-atomic particles. They are different in that electrons are virtually weightless, while protons have measurable weight. Electrons orbit the nucleus of an atom, attracted to the positively charged protons inside the same nucleus.
Additionally, what are the similarities between electrons and neutrons? Neutron is a building block of an atomic nucleus while electrons create charged shells around nuclei so are responsible for the chemical atoms properties.
Also to know, what is the main difference between protons and neutrons quizlet?
Terms in this set (28) What is the difference between protons, neutrons, and electrons? protons are positive, neutrons are neutral, and electrons are smaller and negative.
Why is proton positively charged?
A proton has positive charge of 1, that is, equal but opposite to the charge of an electron. The charge is believed to be from the charge of the quarks that make up the nucleons (protons and neutrons).
Related Question AnswersWhat is the relationship between protons and neutrons?
Protons are bound together in an atom's nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they're neutral). Like protons, neutrons are bound into the atom's nucleus as a result of the strong nuclear force.Where are electrons found?
Electrons are the smallest of the three particles that make up atoms. Electrons are found in shells or orbitals that surround the nucleus of an atom. Protons and neutrons are found in the nucleus. They group together in the center of the atom.What is the charge of a proton?
+1What are protons made of?
Along with neutrons, protons make up the nucleus, held together by the strong force. The proton is a baryon and is considered to be composed of two up quarks and one down quark.How do electrons work?
Electrons are the negatively charged particles of atom. Together, all of the electrons of an atom create a negative charge that balances the positive charge of the protons in the atomic nucleus. Electrons are extremely small compared to all of the other parts of the atom.What is a neutrons charge?
A neutron, like the name implies, is neutral with no net charge. The charge is believed to be from the charge of the quarks that make up the nucleons (protons and neutrons). A neutron is made up of two Down quarks with a negative 1/3 charge each and one Up quark with a positive 2/3 charge.Do neutrons have a charge?
Neutrons and protons are found in the nucleus of an atom. Unlike protons, which have a positive charge, or electrons, which have a negative charge, neutrons have zero charge which means they are neutral particles. A neutron is made of two down quarks and one up quark.Whats is an isotope?
An isotope is any form of a chemical element that has the same number of protons in the nucleus, or the same atomic number, but has a different number of neutrons in the nucleus. Isotopes have the same number of protons but differ in their mass number. Their nuclei contain different numbers of neutrons.What do electrons and protons have in common?
Electrons surround the nucleus. Protons have a positive charge. Electrons have a negative charge. The charge on the proton and electron are exactly the same size but opposite.What is the main difference between protons and neutrons Brainly?
Protons have an electrical charge, but neutrons have no charge. D. Protons make up the nucleus, but neutrons orbit around the nucleus.What happens when two or more atoms react?
What happens when two or more atoms react? Ionic bonds form when electrons are transferred from one atom to another. Ionic compounds are rigid solids with high melting and boiling points and are poor conductors of electricity in their solid states. Covalent bonds involve the sharing of electrons.What are the two parts of an atom?
Number of Protons in an Uncharged Atom The two main components of an atom are the nucleus and the cloud of electrons. The nucleus contains positively charged and neutral subatomic particles, whereas the cloud of electrons contains tiny negatively charged particles.How do you find protons neutrons and electrons quizlet?
Terms in this set (7)- Atomic Number. The same as the number of protons is the same as the number of electrons.
- Atomic Mass. number of protons plus number of neutrons.
- Number of Neutrons. Mass number minus atomic number.
- Atomic Number. number in corner.
- Atomic Mass. number under atomic symbol.
- Atomic Symbol.
- Name of Element.