How are molecules bonded? | ContextResponse.com
.
Then, how are molecules bonded together?
When atoms join together to form molecules, they are held together by chemical bonds. These bonds form as a result of the sharing or exchange of electrons between the atoms. It is only the electrons in the outermost shell that ever get involved in bonding.
Beside above, what are the 4 types of bonds? 4 Types of Chemical Bonds
- 1Ionic bond. Ionic bonding involves a transfer of an electron, so one atom gains an electron while one atom loses an electron.
- 2Covalent bond. The most common bond in organic molecules, a covalent bond involves the sharing of electrons between two atoms.
- 3Polar bond.
Also Know, are all molecules covalently bonded?
Every combination of atoms is a molecule. A compound is a molecule made of atoms from different elements. All compounds are molecules, but not all molecules are compounds. Atoms that share electrons in a chemical bond have covalent bonds.
Are ionic bonds molecules?
Ionic compounds are made up of molecules as well. Ionic compounds have all the characteristics of a compound. The bond that holds the atoms together in it is ionic bond; electrostatic force of attraction. Ionic compounds have 2 or more ions joined together by means of ionic bond into molecules.
Related Question AnswersWhich is strongest bond?
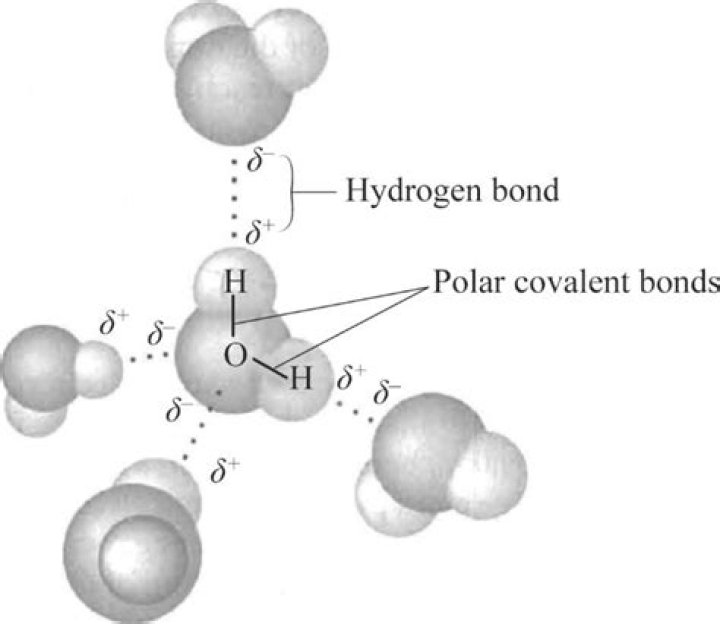
The strongest bond ever is the C-O bond in the carbon monoxide molecule. The second strongest (not much weaker than that) is the triple bond in , which is why it is such a trouble to make nitrogen react with anything.Is water ionic or covalent?
Answer and Explanation: Water is made up of covalent bonds, rather than ionic bonds. The hydrogen atoms share electrons with the oxygen atom, which makes it covalent.Why does chemical bonding occur?
A chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds. The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bonds.Why is NaCl not a molecule?
Molecules have molecular bonds. Something like table salt (NaCl) is a compound because it is made from more than one kind of element (sodium and chlorine), but it is not a molecule because the bond that holds NaCl together is an ionic bond. If you like, you can say that sodium chloride is an ionic compound.How do van der Waals forces hold molecules together?
Van der Waals forces form electrostatic bonds between molecules. Intermolecular bonds including Van der Waals bonds hold the molecules together in liquids and solids and are responsible for phenomena such as the surface tension in liquids and crystals in solids.Why do atoms bond together?
Atoms form chemical bonds with other atoms when there's an electrostatic attraction between them. This attraction results from the properties and characteristics of the atoms' outermost electrons, which are known as valence electrons.What properties do covalently bonded substances have?
Properties of covalent compounds include:- Low boiling points and melting points.
- Various colors.
- Poor conductors of heat and electricity.
- Brittle solids.
What are 3 types of covalent bonds?
The three types as mentioned in the other answers are polar covalent, nonpolar covalent, and coordinate covalent. The first, polar covalent, is formed between two nonmetals that have a difference in electronegativity. They share their electron density unevenly.What atoms are covalently bonded?
Each of the covalent bonds contains two electrons, one from a hydrogen atom and one from the oxygen atom. Both atoms share the electrons. A hydrogen molecule, H2, consists of two hydrogen atoms joined by a covalent bond. Each hydrogen atom needs two electrons to achieve a stable outer electron shell.What are examples of covalent bonds?
Examples of Covalent Bond:- Water. An example is water. Water consists of a covalent bond containing hydrogen and oxygen bonding together to make H2O.
- Diamonds. A diamond is an example of Giant Covalent bond of carbon. A diamond has a giant molecular structure.
- Vulcanized rubber. Another example is vulcanized rubber.