How are chemical energy and nuclear energy alike?
.
Similarly one may ask, what are the similarities between chemical and nuclear reactions?
(1) Nuclear reactions involve a change in anatom's nucleus, usually producing a different element. Chemicalreactions, on the other hand, involve only a rearrangement ofelectrons and do not involve changes in the nuclei.
Likewise, what do fission and fusion have in common? Both fission and fusion are nuclear reactionsthat produce energy, but the applications are not the same.Fission is the splitting of a heavy, unstable nucleus intotwo lighter nuclei, and fusion is the process where twolight nuclei combine together releasing vast amounts ofenergy.
Likewise, people ask, what is a nuclear reaction in chemistry?
In nuclear physics and nuclear chemistry,a nuclear reaction is semantically considered to be theprocess in which two nuclei, or else a nucleus of an atom and asubatomic particle (such as a proton, neutron, or high energyelectron) from outside the atom, collide to produce one or morenuclides that are different from
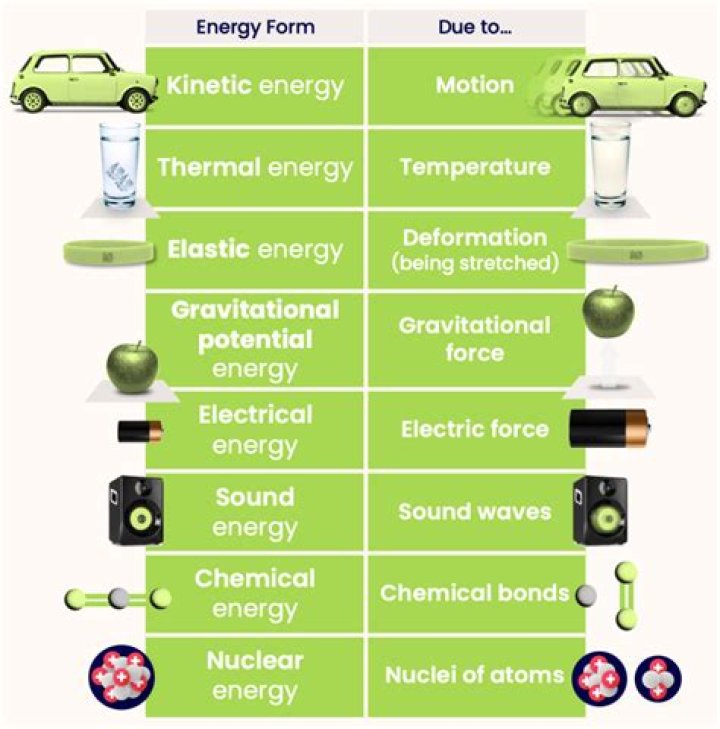
What are the 3 types of energy?
These are the two basic forms of energy. Thedifferent types of energy include thermal energy,radiant energy, chemical energy, nuclearenergy, electrical energy, motion energy,sound energy, elastic energy and gravitationalenergy.
Related Question AnswersWhat are the two types of nuclear reactions?
There are two main types of nuclear reactions,fission and fusion. Nuclear fission is whenthe nucleus of a large atom is split into two or morefragments.Is burning a chemical or physical change?
A chemical change (chemical reaction) is achange of materials into other, new materials with differentproperties, and one or more new substances are formed.Burning of wood is a chemical change as newsubstances which cannot be changed back (e.g. carbon dioxide) areformed.Is Fusion a chemical reaction?
Fusion is not a chemical reaction. It is anuclear reaction. In chemical reactions nuclei don'tchange.What is the key difference between chemical and nuclear reactions?
Chemical reactions involve electronrearrangements. Nuclear reactions involve changes to thenucleus. Explanation; -Nuclear reactions involve a changein an atom's nucleus, usually producing a differentelement ,along with the emission of radiations like α, βand γ etc rays.Which of the chemical reactions is an example of nuclear fusion?
Fusion joins atomic nuclei together. Theelement formed has more neutrons or more protons than that of thestarting material. For example, hydrogen and hydrogen canfuse to form helium. Fission occurs naturally onEarth.What are the three main types of nuclear radiation?
Three types of radioation - Alpha, Beta, Gamma.There are three primary types of radiation: Alpha - these are fastmoving helium atoms. They have high energy, typically in the MeVrange, but due to their large mass, they are stopped by just a fewinches of air, or a piece of paper.How do chemical reactions release energy?
Chemical reactions often involve changes inenergy due to the breaking and formation of bonds.Reactions in which energy is released areexothermic reactions, while those that take in heatenergy are endothermic.Is nuclear fission a chemical reaction?
In nuclear physics and nuclear chemistry,nuclear fission is a nuclear reaction or aradioactive decay process in which the nucleus of an atom splitsinto 2 smaller, lighter nuclei. Fission is a form ofnuclear transmutation because the resulting fragments arenot the same element as the original atom.What are the 4 types of nuclear reactions?
The four main reaction types that will be covered in thisunit are:- Fission.
- Fusion.
- Nuclear Decay.
- Transmutation.