Pour about 1 inch of vinegar into your 2-liter soda bottle using a funnel. Add 2 tablespoons of baking soda into the soda bottle slowly using your funnel. The baking soda and vinegar will fizz. The gas being given off is carbon dioxide..
People also ask, does vinegar and baking soda create co2?
The reaction between baking soda (sodium bicarbonate) and vinegar (dilute acetic acid) generates carbon dioxide gas, which is used in chemical volcanoes and other projects. Here is a look at the reaction between baking soda and vinegar and the equation for the reaction.
Similarly, how does baking soda and vinegar make carbon dioxide? Combine vinegar and baking soda to create a chemical reaction. Carbon dioxide (CO2) gas is formed during this chemical reaction. We can see the evidence of the CO2 in the bubbles.
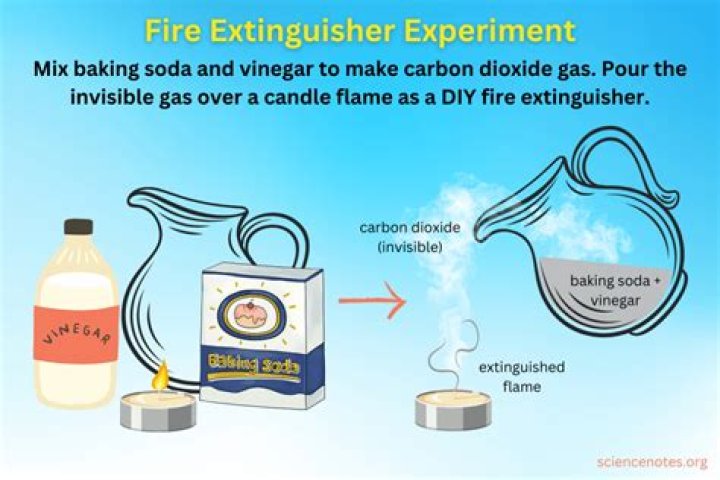
Put out that fire!
- Large liquid measuring cup with a pour spout.
- ½ cup Vinegar (any kind will work)
- 1 Tablespoon Baking Soda.
- Candles.
- Matches.
In this way, how much co2 does vinegar and baking soda produce?
So assuming you take the same proportions of the reactants as their molecular weight is , 44 grams of carbon dioxide will be produced which is equivalent Acetic acid present in vinegar will readily react with baking soda (sodium bicarbonate) to form sodium acetate with the effervescence of carbon dioxide.
Is it harmful to mix vinegar and baking soda?
Baking Soda + Vinegar "When you put them together you get mostly water and sodium acetate. But really, just mostly water." Plus, vinegar causes baking soda to foam up. If stored in a closed container, the mixture can explode.
Related Question Answers
Can baking soda damage your kidneys?
A daily dose of sodium bicarbonate -- baking soda, already used for baking, cleaning, acid indigestion, sunburn and more -- slows the decline of kidney function in some patients with advanced chronic kidney disease, reports an upcoming study.What combination of vinegar and baking soda creates the biggest eruption?
Adding vinegar to baking soda gives you an immediate reaction. Adding baking soda to vinegar, the reaction is delayed, but then fizzes the same amount. More vinegar is better. A 12 to 1 ratio of vinegar to baking soda caused a fizzing explosion!What can I clean with baking soda?
Cleaning with Baking Soda - Spruce up the shower. On a wet microfiber cloth, sprinkle a small amount of baking soda and a couple drops of dishwashing liquid.
- Cleaning drains with baking soda.
- All-purpose scrubber.
- Grout cleaner.
- Bust up grease.
- Mix up a miracle cure.
- Easily clean an oven with baking soda.
- Sink cleaner.
What happens when you mix baking soda and water?
The water that contains baking soda doesn't bubble because it is an alkali, the chemical opposite of an acid. When an alkali combines with an acid, it forms carbon dioxide. When you add baking powder to water or milk, the alkali and the acid react with one another and produce carbon dioxide – the bubbles.What are the products of vinegar and baking soda?
The products of mixing baking soda and vinegar are carbonic acid and the salt sodium acetate. Most of that carbonic acid immediately decomposes to form water and carbon dioxide gas (CO2) which accounts for all the bubbling. You are largely left with a solution of sodium acetate.Do vinegar and baking soda neutralize each other?
When combined, vinegar (an acid) and baking soda (a base) fizz (the reaction) and cancel each other out, which is an awesome science experiment for the kiddos. This fizz is caused by a chemical reaction, resulting in basically salty water which neutralizes the cleaning power of both ingredients.What is baking soda and vinegar used for?
Baking soda, or sodium bicarbonate, is a mild alkali that not only cleans but also deodorizes and is a gentle abrasive. Vinegar is an acid that is an effective, non-toxic alternative to popular cleaners, including bleach. It deodorizes and is often used for the removal of stains, mold, and rust.What is the limiting reagent in baking soda and vinegar?
One underlying assumption is that the baking soda is the only limiting reactant. In other words, there is essentially an unlimited supply of acetic acid in the vinegar bottle, and the reaction output is only dictated by the amount of baking soda you add – every mole added results in a mole of carbon dioxide produced.What happens when baking soda reacts with vinegar?
Acetic acid present in vinegar will readily react with baking soda (sodium bicarbonate) to form sodium acetate with the effervescence of carbon dioxide. The equation for the reaction is: CH3COOH + NaHCO3 = CH3COONa + CO2 + H2O.Could you just keep adding more and more baking soda to the same amount of vinegar to get more carbon dioxide?
Could you just keep adding more and more baking soda to the same amount of vinegar to get more carbon dioxide? No. This might work for a while, as long as there is extra vinegar, but eventually there would be no atoms left of vinegar to react with the extra baking soda, so no more carbon dioxide would be produced.What is the mole ratio of the sodium bicarbonate to acetic acid?
Mole ratio of sodium bicarbonate to acetic acid. there is a mass of . 08g of NaHCO3 and . 00095 moles of it.How long will yeast and sugar produce co2?
More yeast will result in stronger CO2 production, but will exhaust the sugar quicker. Using 1/4 teaspoon of yeast and 2 cups of sugar will result in CO2 production for about 4 to 5 weeks.Is it safe to mix vinegar and Dawn dish soap?
Yes, but I would recommend if you're going to use them both (for washing dishes) then I would suggest washing with soap and then dipping into vinegar water as a “rinse” because the soap will get grease, oil, etc off and then the vinegar would kill most germs. But there's no dangerous byproduct of mixing the two.What is the benefit of baking soda?
Sodium bicarbonate in baking soda neutralises the stomach acid and relieves the many symptoms of acid reflux and other stomach related issues. It is known to be a natural alkalising agent. This non-toxic substance is used to reduce the effect of acid and inflammation in the body and treat most ailments.Is it safe to mix hydrogen peroxide and baking soda?
Hydrogen peroxide is great at whitening and brightening many items, such as stained tiles, dirty grout and even fingernails. For the latter, combine one part hydrogen peroxide with two parts baking soda and rub the paste on your nails. Let sit for two or three minutes, then rinse away.