Does glycogen have a reducing end?
.
Furthermore, does glycogen molecule have a reducing end?
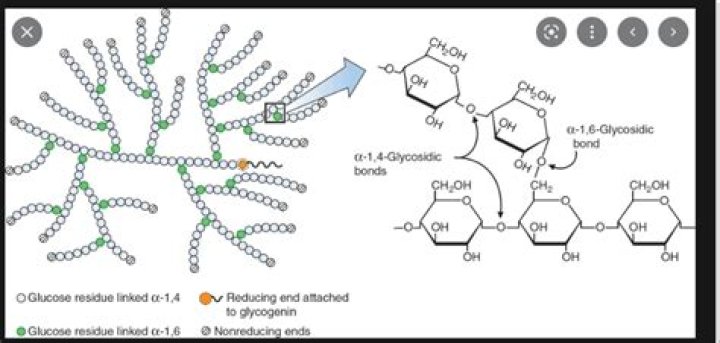
In glycogen, about 10% of the glucose units are branched (Lehninger. p304-305). The molecule has one reducing end (right hand end of chain) but many non-reducing ends (left hand ends) due to the branches.
Also Know, how long do glycogen stores last? Glycogen is both made and stored directly in the liver. When insulin goes up, the body stores food energy as glycogen. When insulin falls, as with fasting, the body breaks glycogen back down into glucose. Liver glycogen lasts approximately 24 hours without eating.
does lactose have a reducing end?
Lactose is a reducing sugar; the β -D-glucose residue has a free anomeric carbon, and therefore the glucose residue is on the reducing end. Lactose is hydrolyzed to glucose and galactose in the intestine by the enzyme lactase. People who are lactose-intolerant lack this enzyme.
How many reducing ends and non reducing ends are found in glycogen and starch?
a) in a molecule of glycogen, there is only one reducing end but the number of non-reducing ends depends on the number of α→6 glycosidic linkages or the branches.
Related Question AnswersHow many reducing ends are in one glycogen?
In glycogen, about 10% of the glucose units are branched (Lehninger. p304-305). The molecule has one reducing end (right hand end of chain) but many non-reducing ends (left hand ends) due to the branches. The nonreducing ends are the locations of all glucose additions or removals.What enzyme breaks down glycogen?
Glycogen phosphorylaseWhat is non reducing end of glycogen?
e.g glycogen has alpha 1,4 linkage with branches having alpha 1,6 linkage. the terminal carbons have no free aldehyde or ketone group, hence called as non-reducing ends, whereas, the carbon 1 of first glucose (present in the core) is reducing end as it has free aldehyde group.Where is glycogen stored?
liverIs Sucrose a Ketose?
Because sucrose is a complex disaccharide, it is not classified as either an aldose or a ketone. Instead, it is a compound that contains both. glucose is aldose sugar and fructose is a ketose sugar. non reducing sugars: do not have an OH group attached to the anomeric carbon so they cannot reduce other compounds.Is dextrose a reducing sugar?
Dextrose is a monosaccharide and a reducing sugar. Starch is a polysaccharide, it is made up of about 10,000 dextrose units. The way in which the units are linked leaves only one at the end of the chain with a reducing capability so starch is not regarded as a reducing compound.Is starch a non reducing sugar?
Starch and Cellulose are polysaccharides. They are comprised of multiple units of glucose linked together. The glucose in starch and cellulose does not contain a free aldehyde group and hence, starch and cellulose do not act as reducing sugars. Hence, sucrose is also not a reducing sugar.Is lactose a reducing sugar?
The common dietary monosaccharides galactose, glucose and fructose are all reducing sugars. Reducing disaccharides like lactose and maltose have only one of their two anomeric carbons involved in the glycosidic bond, while the other is free and can convert to an open-chain form with an aldehyde group.What is a nonreducing end?
The end of the molecule containing the free anomeric carbon is called the reducing end, and the other end is called the nonreducing end. A reducing end of a carbohydrate is a carbon atom that can be in equilibrium with the open-chain aldehyde or keto form.What does Benedict's reagent contain?
Benedict's reagent (often called Benedict's qualitative solution or Benedict's solution) is a chemical reagent and complex mixture of sodium carbonate, sodium citrate and copper(II) sulfate pentahydrate. It is often used in place of Fehling's solution to detect the presence of reducing sugars.What are reducing ends?
In the instance of disaccharides, structures that possess one free unsubstituted anomeric carbon atom are reducing sugars. The end of the molecule containing the free anomeric carbon is called the reducing end, and the other end is called the nonreducing end.Why is glycogen a non reducing sugar?
A reducing sugar is any sugar that is capable of acting as a reducing agent because it has a free aldehyde group or a free ketone group. Glycogen is not a reducing sugar. The only reducing sugars I know are Glucose, Fructose, Galactose, Maltose and Lactose.Why is maltose important?
Lesson Summary So, maltose is a disaccharide that is made up of two glucose units. It has a slightly sweet taste, but it's most important function is in digestion. Since most carbohydrates are in a form that cannot be absorbed, it is important for these carbohydrates to be broken into smaller pieces.Is Lactose a polar molecule?
There are other components, such as calcium ions, sugars(Lactose), proteins(casein), etc. Water is present in considerable amounts and it is a very polar molecule.Why is it called reducing sugar?
Glucose is called a reducing sugar because it can be oxidized by, and thus reduce, mild oxidizing agents such as Cu or Ag. Glucose is an aldose, which means that its open-chain form contains an aldehyde group. Aldehydes are quite easily oxidized to carboxylic acids. So a mild oxidizing agent will be reduced by glucose.How do you know when glycogen is depleted?
Signs and Symptom of Glycogen Depletion- Decreased Strength and Power.
- Increased Rate of Perceived Exertion.
- Feeling of “Flatness” in Muscle Bellies.
- Increase Weight Loss (Water) Overnight.
- Lack of Recovery.
- Decrease Exercise-Performance During Longer Workouts.



