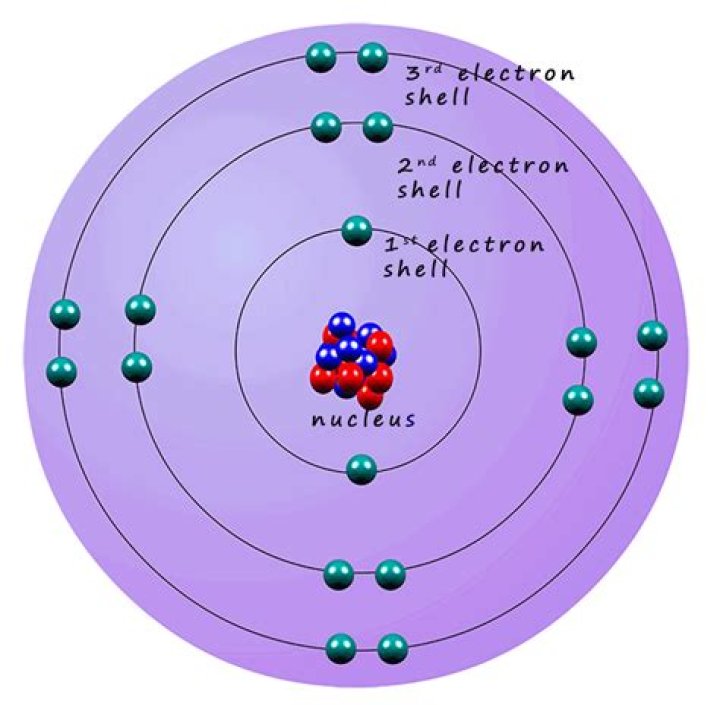
Every atom has at least two electron shells. A full “K" shell has three electrons. A helium atom (atomic number 2) is a "full” atom. A shell must be full before a new shell is started..
In this manner, how many shells can an atom have?
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The general formula is that the nth shell can in principle hold up to 2(n2) electrons.
Subsequently, question is, does oxygen have 2 electron shells? So for the element of OXYGEN, you already know that the atomic number tells you the number of electrons. That means there are 8 electrons in an oxygen atom. Looking at the picture, you can see there are two electrons in shell one and six in shell two. ? More about the history and places to find oxygen.
Hereof, does Shell 2 have 4 electrons?
Lithium (At. No. = 3) has three electrons, two in the K shell and one located farther from the nucleus in the L shell.
Electrons.
| Atomic Number | 4 |
| Element | Beryllium (Be) |
| Energy Levels or "shells" | 2 |
| 2 |
Do all atoms have the same mass?
REMEMBER: Atoms of the same chemical element do not always have the same mass because, although the number of protons in the nucleus is the same for all atoms of the same element, the number of neutrons is not. Most elements as they occur naturally on earth are mixtures of several isotopes.
Related Question Answers
How many electrons can 2s hold?
The 2s subshell holds a maximum of 2 electrons, and the 2p subshell holds a maximum of 6 electrons. Because lithium's final electron goes into the 2s subshell, we write the electron configuration of a lithium atom as 1s 22s 1. The next largest atom, beryllium, has 4 electrons, so its electron configuration is 1s 22s 2.How many atoms are in a gram?
Each of these quantities contains 6.022×1023 atoms of that particular element. The units for molar mass are grams per mole or g/molg/mol.How is Valency calculated?
The valency of an atom is equal to the number of electrons in the outer shell if that number is four or less. Otherwise, the valency is equal to eight minus the number of electrons in the outer shell. Once you know the number of electrons, you can easily calculate the valency.What is a Subshell?
A subshell is a subdivision of electron shells separated by electron orbitals. Subshells are labelled s, p, d, and f in an electron configuration.Why are shells called KLM?
As it turns out, the K type X-ray is the highest energy X-ray an atom can emit. It is produced when an electron in the innermost shell is knocked free and then recaptured. This innermost shell is now called the K-shell, after the label used for the X-ray. Barkla won the 1917 Nobel Prize for Physics for this work.How can you tell how many shells an element has?
The number of electrons is equal to the atom's atomic number, which is at the top left of the element. For example, assume you want to know how many rings are in the element neon. Neon on the periodic table has an atomic number of 10, so it has 10 electrons. Square the ring number, then multiply the result by two.Why are there only 2 electrons in the first shell?
This first shell has only one subshell (labeled 1s) and can hold a maximum of 2 electrons. This is why there are two elements in the first row of the periodic table (H & He). Because the first shell can only hold a maximum of 2 electrons, the third electron must go into the second shell.How many electrons are in the 4th Shell?
The fourth energy level has 18 electrons. The fourth energy level of the periodic table includes the 4s 3d and 4p orbitals. The 4p orbital holds 6 electrons. There is a 4d orbital with 10 electrons which coincides with the 5th energy level of the periodic table.Why are there only 8 electrons in the outer shell?
The eight- electrons stability of an atom is stemmed from the stability of the noble gases or the elder name inert gases, which had long been known as unreactive or noble. However, this rule is justified for elements of second row in the periodic table, which their outermost- shell capacity is 8 electrons.What are the valence electrons in an atom?
In chemistry and physics, a valence electron is an outer shell electron that is associated with an atom, and that can participate in the formation of a chemical bond if the outer shell is not closed; in a single covalent bond, both atoms in the bond contribute one valence electron in order to form a shared pair.How many electrons can the 5th Shell hold?
The first shell only has the s subshell ? 2 electrons. The third shell has the s, p, and d subshells ? 2 + 6 + 10 = 18 electrons. The fifth shell has the s, p, d, f, and g subshells ? 2 + 6 + 10 + 14 + 18 = 50 electrons.What is Shell in atom?
An electron shell is the outside part of an atom around the atomic nucleus. It is where the electrons are, and is a group of atomic orbitals with the same value of the principal quantum number n. Electron shells have one or more electron subshells, or sublevels.What element has 16 neutrons?
phosphorus
How many valence electrons are in Argon?
8 valence electrons
What is Oxygen's atom called?
Oxygen is a chemical element – a substance that contains only one type of atom. Its official chemical symbol is O, and its atomic number is 8, which means that an oxygen atom has eight protons in its nucleus. Oxygen is normally found as a molecule. It is called dioxygen.What is the Valency of oxygen atom?
The valency of oxygen is -2. This means oxygen needs to gain or share two electrons for stability.How many atoms does nitrogen have?
Nitrogen atoms have seven electrons and 7 protons with five electrons in the outer shell.How many atoms are in h2o?
For H2O, there is one atom of oxygen and two atoms of hydrogen. A molecule can be made of only one type of atom. In its stable molecular form, oxygen exists as two atoms and is written O2. to distinguish it from an atom of oxygen O, or ozone, a molecule of three oxygen atoms, O3.Why does oxygen form a 2 ion?
Oxygen has an electron arrangement of (2, 6) and needs to gain two electrons to fill the n=2 energy level and achieve an octet of electrons in the outermost shell. The oxide ion will have a charge of 2− as a result of gaining two electrons.