For example, benzoic acid is not soluble in water, yet it is soluble in sodium hydroxide solution and in sodium hydrogen carbonate solution because these bases react with benzoic acid to form the water-soluble benzoate ion. The first test to perform on all unknowns is water solubility..
Similarly, you may ask, does benzoic acid dissolve in NaOH?
Benzoic acid is a weak acid, thus it will only partially dissolve in water. While NaOH is a strong base, it will attack the weak acid and form a salt. The solubility of benzoic acid in an aqueous NaOH solution is simply the solubility of sodium benzoate in water, a bit over 60 g of sodium benzoate per 100 ml.
Likewise, does benzoic acid dissolve in sodium bicarbonate? A wash with sodium bicarbonate converts benzoic acid into its more water-soluble sodium benzoate form, extracting it into the aqueous layer (Figure 4.57). Additionally, the sodium bicarbonate neutralizes the catalytic acid in this reaction.
Accordingly, is sodium benzoate soluble in NaHCO3?
It is water soluble so is also soluble in aqueous solutions of NaOH or NaHCO3.
What happens when benzoic acid reacts with NaHCO3?
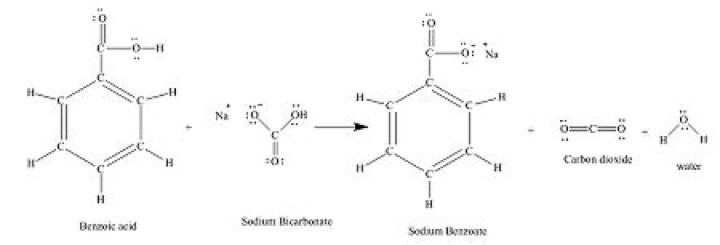
II: Interaction of sodium bicarbonate with substituted benzoic acids in the presence of moisture. The reaction products are carbon dioxide, water, and the sodium salt of the acid. The kinetic rate-determining step for this reaction is the dehydration of carbonic acid.
Related Question Answers
Why benzoic acid is insoluble in HCl?
Thus, benzoic acid is insoluble in cold water. The abundant OH- ions will react fully with benzoic acid in acid-base reaction to form water and benzoate ion, which now is soluble in water due to strong ion-dipole forces of attraction. In acidic medium, benzoic acid becomes insoluble.Is benzoic acid polar or nonpolar?
The primary reason benzoic acid dissolves only slightly in cold water is that, even though the carboxylic acid group is polar, the bulk of the benzoic acid molecule is non-polar (water is polar). It is only the carboxylic group that is polar.Does benzoic acid dissolve in HCl?
Benzoic acid was found to be soluble in water and 1.0 M NaOH, however upon addition of 6.0 M HCl to this solution, benzoic acid became insoluble. Benzoic acid was also insoluble in 1.0 M HCl.What does NaOH do to benzoic acid?
Adding NaOH will neutralize the benzoic acid producing the benzoate ion, which now goes into the aqueous layer, leaving other other two organic compounds in the ether.Does benzoic acid react with NaOH?
Due to its acidic nature, benzoic acid can undergo a reaction with NaOH as follows, resulting in the carboxylate salt sodium benzoate.Is benzamide soluble in NaOH?
Benzamide is a white solid with the chemical formula of C6H5C(O)NH2. It is the simplest amide derivative of benzoic acid. It is slightly soluble in water, and soluble in many organic solvents.Why is NaOH used in extraction?
This phenomenon will often be observed if sodium bicarbonate is used for the extraction in order to neutralize or remove acidic compounds. The reaction affords carbon dioxide (CO2), which is a gas at ambient temperature. Pressure builds up that pushes some of the gas and the liquid out.Are amines soluble in NaOH?
Small, polar organic compounds such as alcohols, aldehydes, ketones, amines, carboxylic acids, and a few phenols are soluble in water. Water-insoluble compounds are first tested with 5% sodium hydroxide (NaOH). Sodium hydroxide is a strong base that ionizes strong or weak (Figure 2 Solubility flowchart) acids.What is soluble in NaOH?
Water Ethanol MethanolHow do you dissolve sodium benzoate?
Sodium benzoate is supplied as a white powder or flake. During use it is mixed dry in bulk liquids where it promptly dissolves. Approximately 1.75 oz (50 g) will readily dissolve in 3 fl oz (100 ml) of water. In contrast, benzoic acid has a significantly lower water solubility profile.Is Sodium Benzoate more polar than benzoic acid?
Sodium benzoate is not a molecule. The benzoic acid molecule is polar because the oxygen atoms, which are more electronegative than carbon and hydrogen, are concentrated on one side of the molecules. Strictly speaking, only symmetrical molecules are non-polar.Does sodium benzoate dissolve in ether?
In general, most neutral, organic compounds will dissolve better in ether than in water. Benzoic acid dissolves better in ether than in water, but sodium benzoate dissolves better in water than in ether.Why does sodium benzoate dissolve in NaOH?
Sodium benzoate will disolve in both the HCl and NaOH aqueous solutions since it is a salt, and salts dissolve in aqueous solutions. Likewise, it will dissolve when added to water to make a sodium benzoate aqueous solutionsolution.Is alcohol soluble in NaOH?
Pure sodium hydroxide is a colorless crystalline solid that melts at 318 °C (604 °F) without decomposition, and with a boiling point of 1,388 °C (2,530 °F). It is highly soluble in water, with a lower solubility in polar solvents such as ethanol and methanol. NaOH is insoluble in ether and other non-polar solvents.Is NaOH polar or nonpolar?
Usually nonpolar covalent, polar covalent, and ionic. As sodium hydroxide has a metal and nonmetallic mix, we can assume that between the sodium and oxygen there is an ionic interaction. And between the hydrogen and oxygen, it's extreme but not as extreme. This is just very polar.What is sodium benzoate soluble in?
Sodium benzoate is a white hygroscopic compound, with a characteristic smell. It has a solubility in water of 62.7 g/100 ml at standard conditions. Sodium benzoate is also soluble in liquid ammonia and pyridine. It is poorly soluble in most alcohols, like methanol and ethanol.Why is HCl used in extraction?
The basic aqueous solutions are neutralized with concentrated HCl to yield the carboxylic acid and phenol, which, because of their water insolubility, precipitate out. These can be collected, recrystallized and allowed to dry to yield pure carboxylic acid and pure phenol.How do you separate phenol and benzoic acid?
Just dissolve all three compounds in an organic solvent which not miscible with water (those two I have mentioned) in separatory funnel. Use aqueous sodium bicarbonate first to extract. If you use sodium hydroxide, both phenol and benzoic acid will be extracted as both can react with hydroxide.How do you separate benzoic acid?
You will use a chemically active extraction to convert the water insoluble benzoic acid into its water soluble salt by treating the carboxylic acid with base. Finally the benzoic acid will be precipitated by adding strong acid to the carboxylate salt solution.