Can a molecule be both polar and nonpolar?
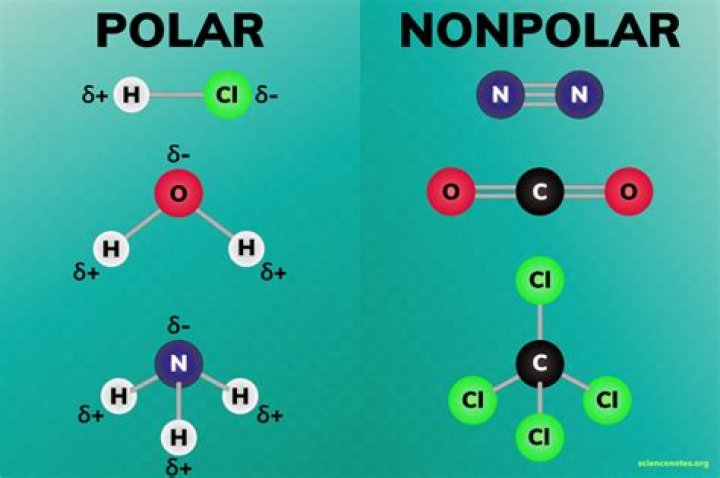
.
Also asked, what is it called when a molecule is both polar and nonpolar?
An amphiphile (from the Greek αμφις, amphis: both and φιλíα, philia: love, friendship) is a chemical compound possessing both hydrophilic (water-loving, polar) and lipophilic (fat-loving) properties. Such a compound is called amphiphilic or amphipathic.
do lone pairs make a molecule polar? Even if all the outer atoms are the same, a molecule with an unsymmetric shape will be (at least slightly) polar. The lone pair or pairs of electrons on the central atom guarantee a nonuniform distribution of electrons. The polarity of the bonds – if they are polar – also contributes to the polarity of the molecule.
Also know, can a nonpolar molecule have polar bonds explain with an example?
Answer and Explanation: YES, a nonpolar molecule can contain polar bonds. The polarity of a molecule depends on the net dipole moment of the molecule.
Is HCL polar or nonpolar?
HCL is a polar molecule as chlorine has a higher electronegativity than the hydrogen. Thus, it attracts electrons to spend more time at its end, giving it a negative charge and hydrogen a positive charge. How do you know if Br2 is polar or nonpolar?
Related Question AnswersIs water non polar?
Water (H2O) is polar because of the bent shape of the molecule. The reason the shape of the molecule isn't linear and nonpolar (e.g., like CO2) is because of the difference in electronegativity between hydrogen and oxygen. The electronegativity value of hydrogen is 2.1, while the electronegativity of oxygen is 3.5.Why is water a polar molecule?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.What determines the polarity of a molecule?
Polarity and Structure of Molecules The shape of a molecule and the polarity of its bonds determine the OVERALL POLARITY of that molecule. If a molecule is completely symmetric, then the dipole moment vectors on each molecule will cancel each other out, making the molecule nonpolar.Is glass polar or nonpolar?
Why is glass polar? - Quora. Regular glass is largely made out of a giant covalent structure of SiO2 molecules - each silicon atom is bonded to two oxygens. Now, oxygen has an electronegativity value of 3.44, which is extremely high. This means that oxygen has a high affinity for attracting the electrons of other atomsWhat is polar or nonpolar?
Polar molecules occur when there is an electronegativity difference between the bonded atoms. Nonpolar molecules occur when electrons are shared equal between atoms of a diatomic molecule or when polar bonds in a larger molecule cancel each other out.Is wax polar or nonpolar?
Paraffin waxes are insoluble because of its non-polar characteristic. All alkanes are non-polar in nature. And since we always say that "like dissolves like", water being a polar molecule, cannot dissolve paraffin waxes.Is ozone a polar molecule?
Ozone is a polar molecule with a dipole moment of 0.53 D.Why is CH3Cl polar?
Since the H is between B and C in terms on electronegativity values, their difference in electronegativity values is so small, the C-H bond is considered nonpolar; thus, no dipole arrow is drawn for the C-H bonds. Because the C-Cl bond is polar, the CH3Cl has a net dipole, so CH3Cl is polar.What does nonpolar mean?
A nonpolar molecule has no separation of charge, so no positive or negative poles are formed. In other words, the electrical charges of nonpolar molecules are evenly distributed across the molecule. Polar molecules tend to dissolve well in water and other polar solvents.What do you mean by polar bonds?
Polar Bond Definition A polar bond is a covalent bond between two atoms where the electrons forming the bond are unequally distributed. This causes the molecule to have a slight electrical dipole moment where one end is slightly positive and the other is slightly negative.What does it mean to be polar?
Polar and Non-Polar Molecules. The arrangement or geometry of the atoms in some molecules is such that one end of the molecule has a positive electrical charge and the other side has a negative charge. If this is the case, the molecule is called a polar molecule, meaning that it has electrical poles.What are some examples of polar molecules?
Examples of polar molecules include:- water - H2O.
- ammonia - NH3.
- sulfur dioxide - SO2.
- hydrogen sulfide - H2S.
- ethanol - C2H6O.



